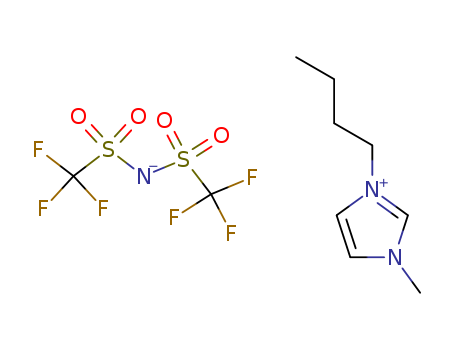
223437-11-4
- Product Name:1-Butyl-1-methylpyrrolidinium Bis(trifluoromethanesulfonyl)imide
- Molecular Formula:C11H20F6N2O4S2
- Purity:99%
- Molecular Weight:422.413
Product Details;
CasNo: 223437-11-4
Molecular Formula: C11H20F6N2O4S2
factory and manufacture 223437-11-4 1-Butyl-1-methylpyrrolidinium Bis(trifluoromethanesulfonyl)imide lonic liquid
- Molecular Formula:C11H20F6N2O4S2
- Molecular Weight:422.413
- Vapor Pressure:0.54mmHg at 25°C
- Melting Point:-6°C
- Refractive Index:1.4216
- Boiling Point:190.5°Cat760mmHg
- Flash Point:69°C
- PSA:85.04000
- Density:g/cm3
- LogP:5.20660
1-BUTYL-1-METHYLPYRROLIDINIUM BIS(TRIFLUOROMETHYLSULFONYL)IMIDE(Cas 223437-11-4) Usage
|
Conductivity |
2.12 mS/cm |
|
General Description |
1-Butyl-1-methylpyrrolidinium bis(trifluoromethylsulfonyl)imide is a room temperature ionic liquid (RTIL) with a wide electrochemical window (5.5V) and high viscosity, making it a useful candidate in electrochemical energy applications. |
InChI:InChI=1/C9H20N.C2F6NO4S2/c1-3-4-7-10(2)8-5-6-9-10;3-1(4,5)14(10,11)9-15(12,13)2(6,7)8/h3-9H2,1-2H3;/q+1;-1
223437-11-4 Relevant articles
Water-based synthesis of hydrophobic ionic liquids for high-energy electrochemical devices
Montanino, Maria,Alessandrini, Fabrizio,Passerini, Stefano,Appetecchi, Giovanni Battista
, p. 124 - 133 (2013)
In this work is described an innovative ...
Fluorescence studies of protein thermostability in ionic liquids
Baker, Sheila N.,Mark McCleskey,Pandey, Siddharth,Baker, Gary A.
, p. 940 - 941 (2004)
Using the single tryptophan residue in t...
Melting behavior and ionic conductivity in hydrophobic ionic liquids
Kunze, Miriam,Montanino, Maria,Appetecchi, Giovanni B.,Jeong, Sangsik,Schoenhoff, Monika,Winter, Martin,Passerini, Stefano
, p. 1776 - 1782 (2010)
Four room-temperature ionic liquids (RTI...
Hybrid ionogel electrolytes for high temperature lithium batteries
Lee, Jin Hong,Lee, Albert S.,Lee, Jong-Chan,Hong, Soon Man,Hwang, Seung Sang,Koo, Chong Min
, p. 2226 - 2233 (2015)
Hybrid ionogels fabricated using 1 M LiT...
Esterification in ionic liquids: The influence of solvent basicity
Wells, Thomas P.,Hallett, Jason P.,Williams, Charlotte K.,Welton, Tom
, p. 5585 - 5588 (2008)
(Chemical Equation Presented) The second...
Heterocyclic bismuth(III) compounds with transannular N→Bi interactions as catalysts for the oxidation of thiophenol to diphenyldisulfide
Toma, Ana M.,Ra?, Ciprian I.,Pavel, Octavian D.,Hardacre, Christopher,Rüffer, Tobias,Lang, Heinrich,Mehring, Michael,Silvestru, Anca,Parvulescu, Vasile I.
, p. 5343 - 5353 (2017)
The reactions between the diorganobismut...
Effects of acetonitrile on electrodeposition of Ni from a hydrophobic ionic liquid
Zhu, Yan-Li,Katayama, Yasushi,Miura, Takashi
, p. 9019 - 9023 (2010)
The effects of addition of acetonitrile ...
Polyethylene glycol-functionalized siloxane hybrid gel polymer electrolytes for lithium ion batteries
Lee, Albert S.,Lee, Jin Hong,Lee, Jong-Chan,Hong, Soon Man,Hwang, Seung Sang,Koo, Chong Min
, p. 3016 - 3020 (2017)
The compatibility of diacrylate terminat...
Cyclic quaternary ammonium ionic liquids with perfluoroalkyltrifluoroborates: Synthesis, characterization, and properties
Zhou, Zhi-Bin,Matsumoto, Hajime,Tatsumi, Kuniaki
, p. 2196 - 2212 (2006)
New cyclic quaternary ammonium salts, co...
Pyrrolidinium imides: A new family of molten salts and conductive plastic crystal phases
MacFarlane,Meakin,Sun,Amini,Forsyth
, p. 4164 - 4170 (1999)
A new family of molten salts is reported...
Ag(I)/Ag electrode reaction in amide-type room-temperature ionic liquids
Serizawa, Nobuyuki,Katayama, Yasushi,Miura, Takashi
, p. 346 - 351 (2010)
Ag(I)/Ag electrode reaction was investig...
Recombination of lophyl radicals in pyrrolidinium-based ionic liquids
Berdzinski, Stefan,Horst, Joachim,Strassburg, Petra,Strehmel, Veronika
, p. 1899 - 1908 (2013)
The recombination of photolytically gene...
Solubilizing and Stabilizing Proteins in Anhydrous Ionic Liquids through Formation of Protein-Polymer Surfactant Nanoconstructs
Brogan, Alex P. S.,Hallett, Jason P.
, p. 4494 - 4501 (2016)
Nonaqueous biocatalysis is rapidly becom...
Electrodeposition of palladium from palladium(II) acetylacetonate in an amide-type ionic liquid
Yoshii, Kazuki,Oshino, Yosuke,Tachikawa, Naoki,Toshima, Kazunobu,Katayama, Yasushi
, p. 21 - 24 (2015)
The electrodeposition of palladium from ...
Physical-Chemical Characterization of Binary Mixtures of 1-Butyl-1-methylpyrrolidinium Bis{(trifluoromethyl)sulfonyl}imide and Aliphatic Nitrile Solvents as Potential Electrolytes for Electrochemical Energy Storage Applications
Neale, Alex R.,Schütter, Christoph,Wilde, Patrick,Goodrich, Peter,Hardacre, Christopher,Passerini, Stefano,Balducci, Andrea,Jacquemin, Johan
, p. 376 - 390 (2017)
In the scope of improving the energy and...
Facile, high-yielding preparation of pyrrolidinium, piperidinium, morpholinium and 2,3-dihydro-1H-isoindolinium salts and ionic liquids from secondary amines
Ward, Antony J.,Masters, Anthony F.,Maschmeyer, Thomas
, p. 23327 - 23337 (2014)
High yield and purity heterocyclic ionic...
Electrochemical Preparation of Platinum Nanoparticles from Bis(acetylacetonato)platinum(II) in Some Aprotic Amide-type Ionic Liquids
Sultana, Sharmin,Tachikawa, Naoki,Yoshii, Kazuki,Toshima, Kazunobu,Magagnin, Luca,Katayama, Yasushi
, p. 263 - 270 (2017)
Electrode reaction of bis(acetylacetonat...
Hybrid ionogel electrolytes derived from polyhedral oligomeric silsesquioxane for lithium ion batteries
Lee, Jin Hong,Lee, Albert S.,Lee, Jong-Chan,Hong, Soon Man,Hwang, Seung Sang,Koo, Chong Min
, p. 3101 - 3104 (2017)
Inorganic-organic hybrid ionogels fabric...
Does the cation really matter? the effect of modifying an ionic liquid cation on an SN2 process
Tanner, Eden E. L.,Yau, Hon Man,Hawker, Rebecca R.,Croft, Anna K.,Harper, Jason B.
, p. 6170 - 6175 (2013)
The rate of reaction of a Menschutkin pr...
Ultrafast dynamics of pyrrolidinium cation ionic liquids
Shirota, Hideaki,Funston, Alison M.,Wishart, James F.,Castner Jr., Edward W.
, (2005)
We have investigated the ultrafast molec...
Newly designed flow reactor as an original method of synthesis of ionic liquids by ion-exchange reactions
Pawlowska-Zygarowicz, Anna
supporting information, (2021/11/16)
Optimization of chemical reactions is of...
Evaluation of ionic liquids as electrolytes for vanadium redox flow batteries
Bahadori, L.,Boyd, R.,Nockemann, P.,Shafeeyan, M. S.,Warrington, A.
, (2020/08/24)
Non-aqueous redox flow batteries (NARFBs...
Controlling the reactions of 1-bromogalactose acetate in methanol using ionic liquids as co-solvents
Gilbert, Alyssa,Haines, Ronald S.,Harper, Jason B.
supporting information, p. 5442 - 5452 (2020/09/09)
The reactions of an acetobromogalactose ...
Binary Mixtures of Aprotic and Protic Ionic Liquids Demonstrate Synergistic Polarity Effect: An Unusual Observation
Thawarkar, Sachin,Khupse, Nageshwar D.,Kumar, Anil
, p. 210 - 221 (2020/02/25)
In this communication, we demonstrate th...
223437-11-4 Process route
-
-
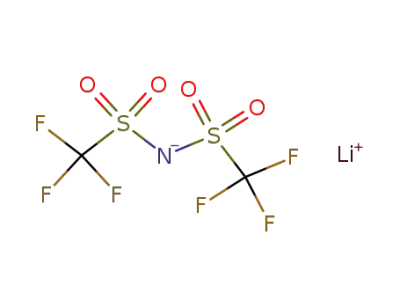
90076-65-6
bis(trifluoromethane)sulfonimide lithium

-
-
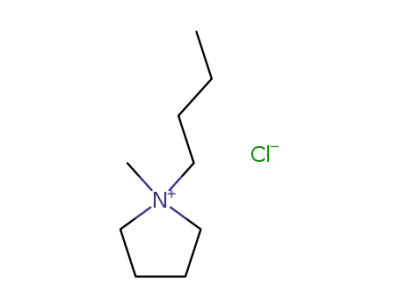
479500-35-1
1-butyl-1-methylpyrrolidinium chloride

-
-
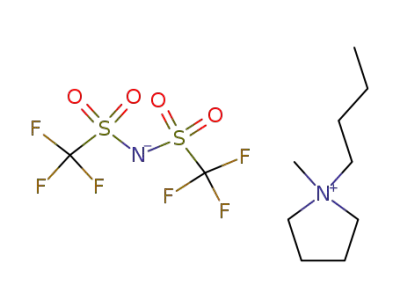
223437-11-4
1-butyl-methylpyrrolidinium bis(trifluoromethylsulfonyl)amide
| Conditions | Yield |
|---|---|
|
In
dichloromethane; water;
Flow reactor;
|
99.3% |
|
In
dichloromethane;
at 120 ℃;
for 24h;
Inert atmosphere;
Schlenk technique;
Glovebox;
|
90% |
|
In
dichloromethane;
at 20 ℃;
for 24h;
|
89% |
|
In
water;
at 20 ℃;
for 17h;
|
88% |
|
In
dichloromethane;
for 48h;
|
87.7% |
|
In
dichloromethane;
for 72h;
|
86% |
|
In
dichloromethane;
for 72h;
|
86% |
|
In
water;
at 20 ℃;
for 24h;
|
86% |
|
In
water;
at 20 ℃;
for 24h;
|
85% |
|
|
|
|
In
dichloromethane; water;
at 20 ℃;
for 24h;
|
|
|
In
water;
for 24h;
|
-

-
90076-65-6
bis(trifluoromethane)sulfonimide lithium

-
-
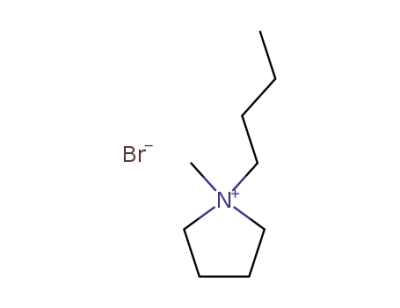
93457-69-3
N-methyl-N-butylpyrrolidinium bromide

-

-
223437-11-4
1-butyl-methylpyrrolidinium bis(trifluoromethylsulfonyl)amide
| Conditions | Yield |
|---|---|
|
In
dichloromethane; water;
|
98% |
|
With
water;
In
dichloromethane;
|
95% |
|
In
dichloromethane;
at 20 ℃;
for 72h;
Inert atmosphere;
|
95.8% |
|
In
dichloromethane; water;
at 20 ℃;
for 19.5h;
|
94% |
|
In
water;
at 20 ℃;
for 24h;
|
93% |
|
In
water;
at 20 ℃;
for 16h;
|
91% |
|
In
water;
at 20 ℃;
for 20h;
|
83% |
|
In
water;
at 20 ℃;
for 20h;
|
82% |
|
In
water;
at 20 ℃;
for 20h;
|
78% |
|
In
water;
at 20 ℃;
|
|
|
In
water;
|
|
|
In
water;
at 20 ℃;
for 3h;
Product distribution / selectivity;
Electrochemical reaction;
|
|
|
In
water;
at 20 ℃;
for 12h;
|
|
|
In
water;
|
|
|
In
water;
at 20 ℃;
|
|
|
In
water;
at 20 ℃;
for 5h;
Inert atmosphere;
|
|
|
In
water;
at 20 ℃;
for 0.0333333 - 0.05h;
Concentration;
Time;
Sealed tube;
Green chemistry;
|
|
|
In
dichloromethane; water;
at 20 ℃;
|
|
|
In
water;
|
|
|
In
water-d2;
|
|
|
In
water;
for 2h;
|
|
|
In
water;
for 2h;
|
|
|
In
dichloromethane; water;
|
|
|
In
water;
at 20 ℃;
|
|
|
In
water;
at 20 ℃;
|
|
|
In
water;
at 20 ℃;
|
223437-11-4 Upstream products
-
56511-17-2
1-n-Butyl-1-methylpyrrolidinium iodide
-
90076-65-6

bis(trifluoromethane)sulfonimide lithium
-
479500-35-1

1-butyl-1-methylpyrrolidinium chloride
-
93457-69-3

N-methyl-N-butylpyrrolidinium bromide
223437-11-4 Downstream products
-
74-90-8

hydrogen cyanide
-
124-38-9
carbon dioxide
-
201230-82-2
carbon monoxide
-
7664-39-3
hydrogen fluoride
Relevant Products
-
1-Butyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide
CAS:174899-83-3
-
2-bromo-3-octylthiophene
CAS:145543-83-5
-
Dimethyl 5-norbornene-2,3-dicarboxylate
CAS:5826-73-3