6825-20-3
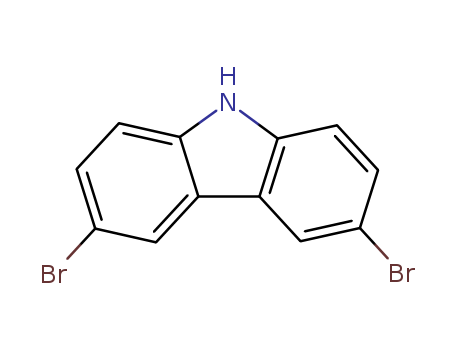
- Product Name:3,6-Dibromocarbazole
- Molecular Formula:C12H7Br2N
- Purity:99%
- Molecular Weight:325.002
Product Details;
CasNo: 6825-20-3
Molecular Formula: C12H7Br2N
Appearance: tan to light green powder
factory and supplier 6825-20-3 3,6-Dibromocarbazole in stock
- Molecular Formula:C12H7Br2N
- Molecular Weight:325.002
- Appearance/Colour:tan to light green powder
- Vapor Pressure:3.58E-08mmHg at 25°C
- Melting Point:204-206 °C(lit.)
- Refractive Index:1.796
- Boiling Point:459 °C at 760 mmHg
- PKA:15.47±0.30(Predicted)
- Flash Point:231.4 °C
- PSA:15.79000
- Density:1.93 g/cm3
- LogP:4.84610
3,6-Dibromocarbazole(Cas 6825-20-3) Usage
|
Synthesis |
3, 6-dibromocarbazole is synthesized with three methods, such as N-bromosuccinimide method, liquid bromine method and silica gel method. But now main method for synthesis is silica gel method. The main raw materials used in the synthesis include carbazole, N-bromosuccinimide, solvent methylene chloride, catalyst silica gel, and the yield could reach 89.5%. |
|
General Description |
Lithiation of 3,6-dibromocarbazole with n-butyllithium has been studied. |
InChI:InChI=1/C12H7Br2N/c13-7-1-3-11-9(5-7)10-6-8(14)2-4-12(10)15-11/h1-6,15H
6825-20-3 Relevant articles
Synthesis and luminescent properties of novel organic luminescent materials based on carbazole derivatives
Duan, Yingxiang,Yang, Yanhua,Cen, Bo,Shen, Kecheng,Tao, Xian,Shen, Yingzhong
, p. 2955 - 2963 (2018)
Four novel host materials C1–C4 based on...
Syntheses, characterizations and electrochromic applications of polymers derived from carbazole containing thiophene rings in side chain with electrochemical and FeCl3 methods
Aydin, Aysel,Kaya, Ismet
, p. 730 - 743 (2013)
1,8-Bis(3,6-di(thiophen-3-yl)-9H-carbazo...
Synthesis and characterization of two carbazole-based alternating copolymers with 4-nitrophenylcyanovinylene pendant groups and their use as electron donors for bulk heterojunction solar cells
Sharma,Singh, Manjeet,Kurchania, Rajnish,Koukaras,Mikroyannidis
, p. 18821 - 18834 (2013)
Two new carbazole-based copolymers PT an...
Synthesis and characterization of red light-emitting electrophosphorescent polymers with different triplet energy main chain
Ma, Zhihua,Ding, Junqiao,Cheng, Yanxiang,Xie, Zhiyuan,Wang, Lixiang,Jing, Xiabin,Wang, Fosong
, p. 2189 - 2197 (2011)
Red light-emitting electrophosphorescent...
Organic dyes containing fused acenes as building blocks: Optical, electrochemical and photovoltaic properties
Gao, Peng,Tsao, Hoi Nok,Teuscher, Joel,Gr?tzel, Michael
, p. 289 - 292 (2018)
Two D-π-A dyes based on fused acenes (ca...
Blue light-emitting polyfluorenes containing dibenzothiophene-S,S-dioxide unit in alkyl side chain
Fang, Yiwen,Liu, Jie,Zhang, Yuhao,Guo, Ting,Huang, Fei,Yang, Wei,Cao, Yong
, p. 1356 - 1366 (2017)
Blue light-emitting polyfluorenes contai...
Hydrogen-Bonded Crystalline Molecular Machines with Ultrafast Rotation and Displacive Phase Transitions
Colin-Molina, Abraham,Jellen, Marcus J.,Rodríguez-Hernández, Joelis,Cifuentes-Quintal, Miguel Eduardo,Barroso, Jorge,Toscano, Rubén A.,Merino, Gabriel,Rodríguez-Molina, Braulio
, p. 11727 - 11733 (2020)
Two new crystalline rotors 1 and 2 assem...
Synthesis and optical characterization of bipod carbazole derivatives
?a?ll, Merve,?i?ek, Baki,Tülek, Remziye,Teke, Ali
, p. 148 - 156 (2020)
In this study, some new biscarbazole der...
Synthesis and characterization of yellow and green light emitting novel polymers containing carbazole and electroactive moieties
Aydin, Aysel,Kaya, Ismet
, p. 104 - 114 (2012)
The compounds 1,5-bis(3,6-di(thiophen-3-...
Preparation and properties of poly[9-hexadecyl-3-phenyl-6-(4-vinylphenyl)- 9H-carbazole]
Bakiev,Shklyaeva,Lunegov,Mokrushin,Abashev
, p. 1313 - 1319 (2014)
Preparation of new carbazole-containing ...
Synthesis and characterization of 1,3,5-triphenylamine derivatives with star-shaped architecture
Brzeczek, Alina,Karon, Krzysztof,Higginbotham, Heather,J?drysiak, Rafa? G.,Lapkowski, Mieczyslaw,Walczak, Krzysztof,Golba, Sylwia
, p. 25 - 32 (2016)
In this work we report the synthesis, el...
Synthesis of 3,6-dibromocarbazole
Polivin, Yu. N.,Karakhanov, R. A.,Kelarev, V. I.,Bratkov, A. A.,Ugrak, B. I.,et al.
, p. 214 (1993)
-
A fluorescent lanthanide-organic framework for highly sensitive detection of nitroaromatic explosives
Mu, Yajuan,Ran, Yungen,Du, Jianlong,Wu, Xueyi,Nie, Weiwei,Zhang, Jianhong,Zhao, Ying,Liu, Huan
, p. 125 - 130 (2017)
A lanthanide-organic framework construct...
Synthesis and spectroelectrochemical characterization of multi-colored novel poly(3,6-dithienylcarbazole) derivatives containing azobenzene and coumarin chromophore units
Yi?it, Deniz,Hacioglu, Serife O.,Güllü, Mustafa,Toppare, Levent
, p. 140 - 152 (2016)
Azobenzene and coumarin units bearing fo...
Architectural design of new conjugated systems carrying donor-π-acceptor groups (carbazole-CF3): Characterizations, optical, photophysical properties and DSSC's applications
Caglar, Mujdat,Caglar, Yasemin,Derince, Betul,Gorgun, Kamuran
, (2021/10/27)
In this study, two new organic dyes cont...
Functionalization of biphenylcarbazole (CBP) with siloxane-hybrid chains for solvent-free liquid materials
Correia, Gabriel,Heinrich, Beno?t,Méry, Stéphane,Mager, Lo?c,Polychronopoulou, Kyriaki,Ribierre, Jean-Charles,Shaya, Janah
, (2021/12/29)
We report herein the synthesis of siloxa...
A Family of Superhelicenes: Easily Tunable, Chiral Nanographenes by Merging Helicity with Planar π Systems
Amsharov, Konstantin Y.,B?nisch, Simon,G?rling, Andreas,Guldi, Dirk M.,Haines, Philipp,Hampel, Frank,Jelfs, Kim E.,Jux, Norbert,Nelson, Jenny,Reger, David,Schmidt, Julia A.,Ullrich, Tobias
supporting information, p. 18073 - 18081 (2021/07/16)
We designed a straightforward synthetic ...
A nitrophenyl-carbazole based push-pull linker as a building block for non-linear optical active coordination polymers: A structural and photophysical study
Fischer, Roland A.,Hauer, Jürgen,Mayer, David C.,P?thig, Alexander,Thyrhaug, Erling,Weish?upl, Sebastian J.
, (2020/12/07)
Non-linear optical effects (NLO) such as...
6825-20-3 Process route
-
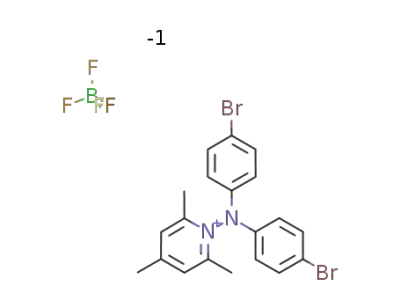
-
N-(4,4′-dibromodiphenylamino)-2,4,6-trimethylpyridinium tetrafluoroborate

-
-
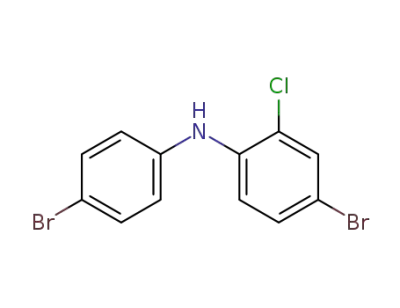
4-bromo-N-(4-bromophenyl)-2-chloroaniline

-
-
6825-20-3
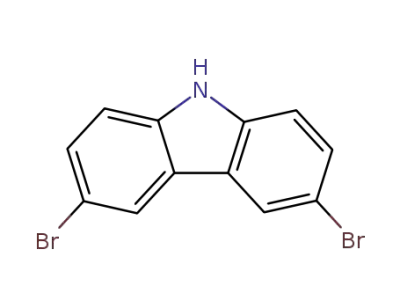
3,6-dibromo-9H-carbazole

-
-
16292-17-4
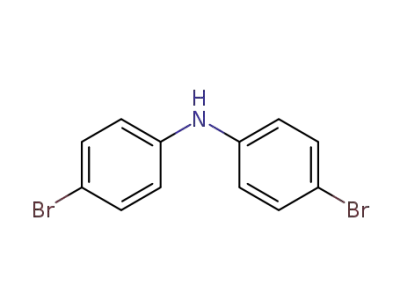
bis(4-bromophenyl)amine
| Conditions | Yield |
|---|---|
|
With
sodium chloride;
In
acetonitrile;
Photolysis;
|
6.8 mg |
-
-
86-74-8,105184-46-1,97960-57-1
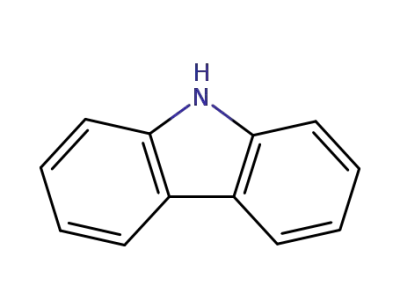
9H-carbazole

-

-
6825-20-3
3,6-dibromo-9H-carbazole
| Conditions | Yield |
|---|---|
|
With
N-Bromosuccinimide;
In
tetrahydrofuran;
at 85 ℃;
for 48h;
|
98% |
|
With
N-Bromosuccinimide;
In
dichloromethane; N,N-dimethyl-formamide;
at 20 ℃;
|
97% |
|
With
N-Bromosuccinimide;
In
N,N-dimethyl-formamide;
at 0 - 20 ℃;
for 2.5h;
|
97% |
|
With
N-Bromosuccinimide;
In
N,N-dimethyl-formamide;
at 0 - 20 ℃;
for 2h;
|
97% |
|
With
1,3-dibromo-5,5-dimethylimidazolidine-2,4-dione;
In
ethanol;
at 20 - 25 ℃;
for 2h;
|
96% |
|
With
N-Bromosuccinimide;
In
N,N-dimethyl-formamide;
at 0 ℃;
for 24h;
|
96% |
|
With
N-Bromosuccinimide;
In
N,N-dimethyl-formamide;
Inert atmosphere;
Schlenk technique;
|
96% |
|
With
N-Bromosuccinimide;
In
ethyl acetate;
at 0 - 20 ℃;
for 5h;
Inert atmosphere;
Schlenk technique;
Darkness;
Sealed tube;
|
95% |
|
With
N-Bromosuccinimide;
In
N,N-dimethyl-formamide;
at 0 ℃;
for 2h;
|
93% |
|
With
N-Bromosuccinimide;
In
N,N-dimethyl-formamide; toluene;
for 0.5h;
Cooling with ice;
Inert atmosphere;
|
92% |
|
With
N-Bromosuccinimide; silica gel;
In
dichloromethane;
at 20 ℃;
for 5h;
|
91% |
|
With
N-Bromosuccinimide;
In
N,N-dimethyl-formamide;
at 0 - 20 ℃;
|
91% |
|
With
N-Bromosuccinimide;
In
dichloromethane; N,N-dimethyl-formamide;
at 20 ℃;
|
90% |
|
With
N-Bromosuccinimide;
In
dichloromethane; N,N-dimethyl-formamide;
at 20 ℃;
|
90% |
|
With
N-Bromosuccinimide;
In
dichloromethane;
for 4h;
|
90% |
|
With
N-Bromosuccinimide;
In
N,N-dimethyl-formamide;
at 0 ℃;
|
90% |
|
With
N-Bromosuccinimide; silica gel;
In
dichloromethane;
at 20 ℃;
for 24h;
|
88% |
|
With
silica gel;
In
dichloromethane;
at 20 ℃;
Darkness;
|
87% |
|
With
N-Bromosuccinimide;
In
N,N-dimethyl-formamide;
at 0 - 20 ℃;
|
86% |
|
With
N-Bromosuccinimide;
In
dichloromethane;
at 20 ℃;
for 6h;
Inert atmosphere;
|
85% |
|
With
N-Bromosuccinimide;
In
N,N-dimethyl-formamide;
at 0 - 20 ℃;
for 6h;
Flash photolysis;
|
85% |
|
With
N-Bromosuccinimide;
In
tetrahydrofuran;
at 20 ℃;
for 4h;
|
83% |
|
With
N-Bromosuccinimide;
In
tetrahydrofuran;
at 20 ℃;
for 4h;
|
83% |
|
With
N-Bromosuccinimide;
In
dichloromethane;
for 12h;
Cooling with ice;
Darkness;
|
83% |
|
With
Oxone; copper(ll) bromide;
In
acetonitrile;
at 20 ℃;
for 5h;
regioselective reaction;
|
82% |
|
With
bromine;
In
chloroform;
at 0 - 20 ℃;
for 13h;
|
82% |
|
With
N-Bromosuccinimide;
In
dichloromethane; N,N-dimethyl-formamide;
at 20 ℃;
|
82% |
|
With
N-Bromosuccinimide;
In
dichloromethane; N,N-dimethyl-formamide;
at 20 ℃;
|
82% |
|
With
N-Bromosuccinimide;
In
dichloromethane;
at 20 ℃;
|
82% |
|
With
N-Bromosuccinimide;
In
N,N-dimethyl-formamide;
at 20 ℃;
|
80% |
|
With
N-Bromosuccinimide; sodium hydride;
In
hexane; N,N-dimethyl-formamide;
at 0 - 20 ℃;
for 2h;
|
80% |
|
With
N-Bromosuccinimide;
In
tetrahydrofuran;
for 4h;
Inert atmosphere;
Cooling with ice;
|
80% |
|
With
N-Bromosuccinimide; silica gel;
In
1,2-dichloro-ethane;
at 20 ℃;
for 18h;
Inert atmosphere;
Darkness;
|
78% |
|
With
N-Bromosuccinimide;
In
tert-butyl methyl ether;
at 100 ℃;
for 12h;
Sealed tube;
|
78% |
|
With
N-Bromosuccinimide; silica gel;
In
dichloromethane;
|
76% |
|
With
N-Bromosuccinimide; silica gel;
In
dichloromethane;
at 20 ℃;
for 24h;
Darkness;
|
75% |
|
With
N-Bromosuccinimide;
In
tetrahydrofuran;
at 40 ℃;
for 20h;
|
75% |
|
With
N-Bromosuccinimide; silica gel;
In
dichloromethane;
at 22 ℃;
for 16h;
Inert atmosphere;
Darkness;
|
74% |
|
With
N-Bromosuccinimide; dibenzoyl peroxide;
In
dichloromethane;
at 20 ℃;
|
73% |
|
With
N-Bromosuccinimide; dibenzoyl peroxide;
In
dichloromethane;
at 20 ℃;
|
73% |
|
With
N-Bromosuccinimide;
In
N,N-dimethyl-formamide;
at 0 - 20 ℃;
for 2h;
|
72% |
|
With
N-Bromosuccinimide; silica gel;
In
dichloromethane;
at 20 ℃;
for 24h;
Darkness;
|
72% |
|
With
N-Bromosuccinimide;
In
N,N-dimethyl-formamide;
for 0.5h;
Cooling with ice;
|
68% |
|
With
hydrogen bromide; dimethyl sulfoxide;
at 80 ℃;
for 10h;
|
67% |
|
With
N-Bromosuccinimide; silica gel;
In
dichloromethane;
at 20 ℃;
for 6h;
|
66% |
|
With
N-Bromosuccinimide;
In
N,N-dimethyl-formamide;
for 2h;
Cooling with ice;
|
65% |
|
With
N-Bromosuccinimide; silica gel;
In
dichloromethane;
at 20 ℃;
for 12h;
Darkness;
|
64% |
|
With
N-Bromosuccinimide; silica gel;
In
dichloromethane;
at 20 ℃;
for 12h;
|
63% |
|
With
N-Bromosuccinimide;
In
dichloromethane; N,N-dimethyl-formamide;
for 3h;
Inert atmosphere;
|
61.5% |
|
With
N-Bromosuccinimide;
In
N,N-dimethyl-formamide;
for 4h;
|
59% |
|
With
N-Bromosuccinimide;
In
N,N-dimethyl-formamide;
at 0 - 20 ℃;
for 2h;
Inert atmosphere;
|
55% |
|
With
bromine; acetic acid;
In
ethanol;
|
52.1% |
|
With
bromine;
In
carbon disulfide;
at 50 ℃;
for 5h;
|
47% |
|
With
N-Bromosuccinimide;
In
N,N-dimethyl-formamide;
at 20 ℃;
Cooling with ice;
|
47% |
|
With
N-Bromosuccinimide;
In
N,N-dimethyl-formamide; toluene;
at 0 - 20 ℃;
for 1.5h;
Inert atmosphere;
Schlenk technique;
|
43% |
|
With
N-Bromosuccinimide;
In
tetrahydrofuran;
at 20 ℃;
for 3.66667h;
|
43.8% |
|
With
potassium bromate; hydrogenchloride; acetic acid; potassium bromide;
|
|
|
With
carbon disulfide; bromine;
|
|
|
With
bromine; acetic anhydride; acetic acid;
|
|
|
With
N-Bromosuccinimide; silica gel;
In
dichloromethane;
at 18 ℃;
for 4.5h;
Yield given;
|
|
|
With
tetra-N-butylammonium tribromide;
In
chloroform;
for 0.5h;
Ambient temperature;
|
100 % Chromat. |
|
With
N-Bromosuccinimide;
In
acetonitrile;
at 20 ℃;
Inert atmosphere;
Cooling with ice;
|
|
|
With
N-Bromosuccinimide;
In
dichloromethane;
|
|
|
With
N-Bromosuccinimide;
In
N,N-dimethyl-formamide;
at 20 ℃;
|
|
|
With
N-Bromosuccinimide;
In
N,N-dimethyl-formamide;
|
|
|
With
bromine; sodium acetate; acetic acid;
at 0 ℃;
|
|
|
With
N-Bromosuccinimide;
In
N,N-dimethyl-formamide;
at 20 ℃;
|
|
|
With
N-Bromosuccinimide; silica gel; potassium carbonate;
In
dichloromethane;
for 24h;
Inert atmosphere;
|
|
|
With
N-Bromosuccinimide;
In
N,N-dimethyl-formamide;
|
|
|
With
N-Bromosuccinimide;
In
N,N-dimethyl-formamide;
|
|
|
With
N-Bromosuccinimide;
In
N,N-dimethyl-formamide; toluene;
at 0 ℃;
for 2h;
Inert atmosphere;
|
|
|
With
N-Bromosuccinimide;
|
|
|
With
N-Bromosuccinimide;
In
tetrachloromethane;
for 1h;
Reflux;
|
|
|
With
N-Bromosuccinimide;
In
N,N-dimethyl-formamide;
at 20 ℃;
|
|
|
With
N-Bromosuccinimide;
In
N,N-dimethyl-formamide; toluene;
at 0 ℃;
|
|
|
With
N-Bromosuccinimide;
In
N,N-dimethyl-formamide;
for 0.5h;
|
|
|
With
N-Bromosuccinimide;
Inert atmosphere;
|
|
|
With
N-Bromosuccinimide; silica gel;
In
dichloromethane;
at 20 ℃;
|
|
|
With
N-Bromosuccinimide; silica gel;
|
|
|
With
N-Bromosuccinimide;
In
chloroform;
at 20 ℃;
|
|
|
With
N-Bromosuccinimide;
In
N,N-dimethyl-formamide; toluene;
|
|
|
With
bromine;
In
carbon disulfide;
|
|
|
With
bromine; acetic anhydride; acetic acid;
|
|
|
With
N-Bromosuccinimide;
In
N,N-dimethyl-formamide;
at 95 ℃;
for 12h;
Schlenk technique;
Inert atmosphere;
|
|
|
With
N-Bromosuccinimide;
In
N,N-dimethyl-formamide;
|
|
|
With
N-Bromosuccinimide;
In
N,N-dimethyl-formamide;
at 20 ℃;
for 1h;
|
|
|
With
N-Bromosuccinimide;
In
dichloromethane; N,N-dimethyl-formamide;
at 25 ℃;
for 25h;
|
3.6 g |
|
With
N-Bromosuccinimide;
In
dichloromethane; N,N-dimethyl-formamide;
at 20 ℃;
for 24h;
|
3.6 g |
|
With
N-Bromosuccinimide;
In
N,N-dimethyl-formamide;
at 0 ℃;
for 3h;
|
|
|
With
N-Bromosuccinimide;
In
tetrahydrofuran; N,N-dimethyl-formamide;
at 40 ℃;
|
|
|
With
N-Bromosuccinimide;
In
toluene;
|
6825-20-3 Upstream products
-
228091-77-8
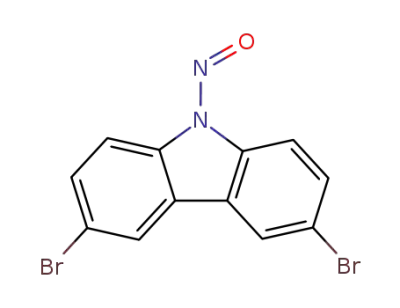
N-nitroso-3,6-dibromocarbazole
-
67-64-1
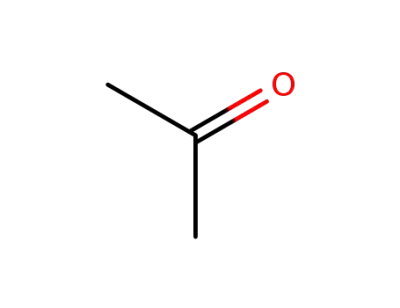
acetone
-
912850-81-8
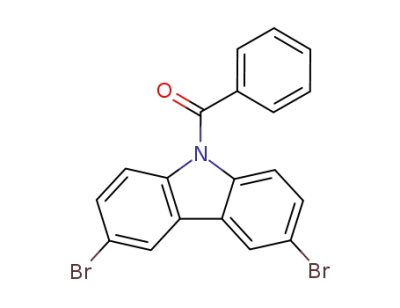
9-benzoyl-3,6-dibromo-carbazole
-
77-48-5
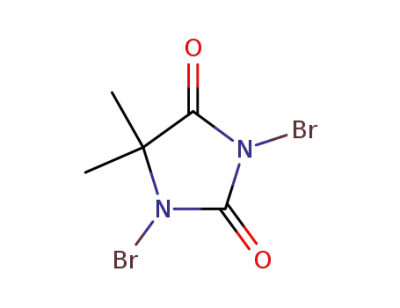
1,3-dibromo-5,5-dimethylimidazolidine-2,4-dione
6825-20-3 Downstream products
-
33255-13-9
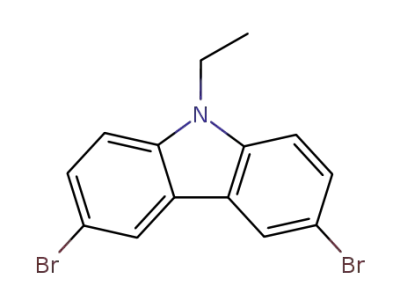
3,6-dibromo-9-ethyl-9H-carbazole
-
38573-63-6
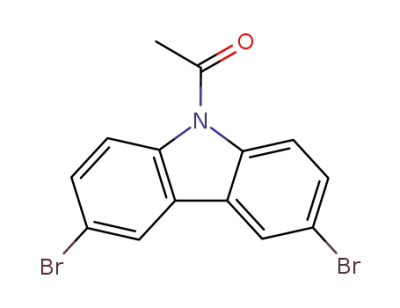
9-acetyl-3,6-dibromocarbazole
-
58246-82-5
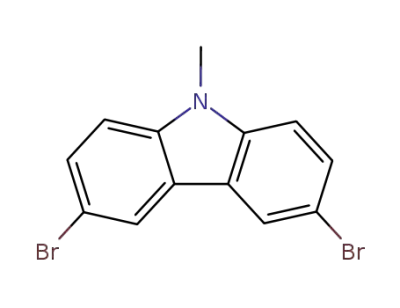
3,6-dibromo-9-methyl-9H-carbazole
-
5416-22-8
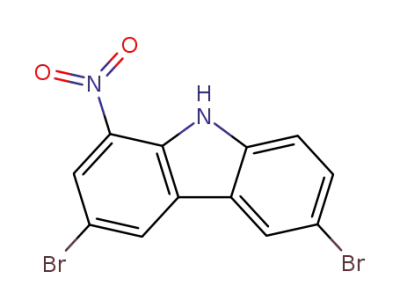
3,6-dibromo-1-nitrocarbazole
Relevant Products
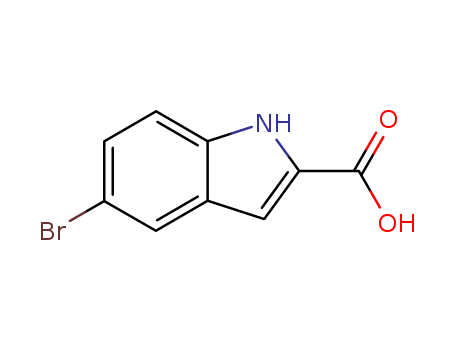
-
5-Bromoindole-2-carboxylic acid
CAS:7254-19-5
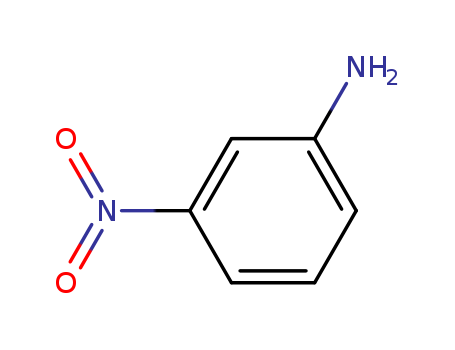
-
3-Nitroaniline
CAS:99-09-2
-
2-Bromo-m-xylene
CAS:576-22-7