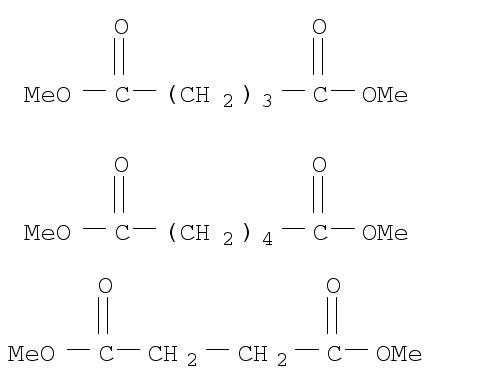119-64-2
- Product Name:1,2,3,4-Tetrahydronaphthalene
- Molecular Formula:C10H12
- Purity:99%
- Molecular Weight:132.205
Product Details;
CasNo: 119-64-2
Molecular Formula: C10H12
Appearance: colourless liquid with a mouldy smell
factory and supplier 119-64-2 1,2,3,4-Tetrahydronaphthalene in stock
- Molecular Formula:C10H12
- Molecular Weight:132.205
- Appearance/Colour:colourless liquid with a mouldy smell
- Vapor Pressure:0.18 mm Hg ( 20 °C)
- Melting Point:-35 °C(lit.)
- Refractive Index:1.5410 - 1.5411
- Boiling Point:210.29 °C at 760 mmHg
- Flash Point:77.222 °C
- PSA:0.00000
- Density:0.97 g/cm3
- LogP:2.56540
1,2,3,4-Tetrahydronaphthalene(Cas 119-64-2) Usage
|
Production Methods |
Tetralin is prepared by the catalytic hydrogenation of naphthalene or during acidic, catalytic hydrocracking of phenanthrene. At 700℃, tetralin yields tars that contain appreciable quantities of 3,4-benzopyrene (172a). |
|
Synthesis Reference(s) |
Journal of the American Chemical Society, 111, p. 314, 1989 DOI: 10.1021/ja00183a048Tetrahedron Letters, 12, p. 1853, 1971 |
|
Air & Water Reactions |
Flammable. |
|
Reactivity Profile |
1,2,3,4-Tetrahydronaphthalene may react vigorously with strong oxidizing agents. May react exothermically with reducing agents to release hydrogen gas. Oxidizes readily in air to form unstable peroxides that may explode spontaneously [Bretherick 1979 p.151-154]. |
|
Hazard |
Irritant to eyes and skin; narcotic in high concentration. |
|
Health Hazard |
Liquid may cause nervous disturbance, green coloration of urine, and skin and eye irritation |
|
Carcinogenicity |
In male and female F344/N and NBR rats exposed to tetralin at concentrations of 0, 30, 60, or 120 ppm, 6 h plus T90 (12 min) per day, 5 days per week for 105 weeks, there were slightly increased incidences of cortical renal tubule adenoma in male rats. The incidence of cortical renal tubule adenomawas also significantly increased in the 120 ppm group. Exposure of male and female B6C3F1 mice to tetralin at concentrations of 0, 30, 60, or 120 ppm, 6 h plus T90 (12 min) per day, 5 days per week for 105 weeks and additional groups of male and female mice to the same concentrations for 12 months led to increased incidence of hemangiosarcoma of the spleen in 120 ppm females (172b). |
|
Purification Methods |
Wash tetralin with successive portions of conc H2SO4 until the acid layer is no longer coloured, then wash it with aqueous 10% Na2CO3, and then distilled water. Dry (CaSO4 or Na2SO4), filter, reflux and fractionally distil it under under reduced pressure from sodium or BaO. It can also be purified by repeated fractional freezing. Bass [J Chem Soc 3498 1964] freed tetralin, purified as above, from naphthalene and other impurities by conversion to ammonium tetralin-6-sulfonate. Concentrated H2SO4 (150mL) is added slowly to stirred tetralin (272mL) which is then heated on a water bath for about 2hours for complete solution. The warm mixture, when poured into aqueous NH4Cl solution (120g in 400mL water), gives a white precipitate which, after filtering off, is crystallised from boiling water, washed with 50% aqueous EtOH and dried at 100o. Evaporation of its boiling aqueous solution on a steam bath removes traces of naphthalene. The pure salt (229g) is mixed with conc H2SO4 (266mL) and steam distilled from an oil bath at 165-170o. An ether extract of the distillate is washed with aqueous Na2SO4, and the ether is evaporated, prior to distilling the tetralin from sodium. Tetralin has also been purified via barium tetralin-6-sulfonate, conversion to the sodium salt and decomposed in 60% H2SO4 using superheated steam. [Beilstein 5 H 491, 5 III 1219, 5 IV 1388.] |
|
General Description |
1,2,3,4-Tetrahydronaphthalene (Tetralin) is a hydrogenated derivative of naphthalene, commonly used as a solvent and hydrogen donor in organic reactions. It participates in transfer hydrogenation processes, such as the selective hydrogenation of polycyclic aromatic hydrocarbons (e.g., anthracene and naphthacene) when catalyzed by molten antimony trichloride, forming dihydro derivatives without complete dehydrogenation to naphthalene. Additionally, Tetralin acts as a hydrogen donor in thermal decomposition reactions, where it facilitates reverse radical disproportionation in certain solvents, leading to varied product distributions. Its utility extends to synthetic applications, including Friedel-Crafts cyclizations, where it contributes to the formation of enantiopure tetrahydronaphthalene derivatives. |
|
Definition |
ChEBI: An ortho-fused bicyclic hydrocarbon that is 1,2,3,4-tetrahydro derivative of naphthalene. |
InChI:InChI=1/C10H12/c1-2-6-10-8-4-3-7-9(10)5-1/h1-2,5-6H,3-4,7-8H2
119-64-2 Relevant articles
Magnetically separable mesoporous silica-supported palladium nanoparticle-catalyzed selective hydrogenation of naphthalene to tetralin
Yang, Yonghui,Xu, Bolian,He, Jie,Shi, Jianjun,Yu, Lei,Fan, Yining
, (2019)
A novel magnetically separable mesoporou...
Cp?Ir-Catalyzed Acceptorless Dehydrogenation of Carbon-Carbon Single Bonds
Ando, Hideaki,Kusumoto, Shuhei,Wu, Weiwei,Nozaki, Kyoko
, p. 2317 - 2322 (2017)
Pentamethylcyclopentadienyl (Cp?) iridiu...
Treatment of naphthols with B(C6F5)3: Formation and characterization of the Lewis acid adducts of their keto isomers
Vagedes, Dominik,Froehlich, Roland,Erker, Gerhard
, p. 3362 - 3365 (1999)
With the strong Lewis acid B(C6F5)3, the...
Hydrogenation of Aromatics in Diesel Fuels on Pt/MCM-41 Catalysts
Corma,Martinez,Martinez-Soria
, p. 480 - 489 (1997)
The hydrogenation activity of Pt support...
Synthesis of Ni–W aromatic hydrocarbon hydrogenation catalysts by the ex situ and in situ decomposition of a precursor based on a dendrimer network
Sizova,Kulikov,Zolotukhina,Serdyukov,Maksimov,Karakhanov
, p. 1107 - 1113 (2016)
A Ni–W precursor supported on a dendrime...
Mechanisms of Decomposition of the Ene Adducts of Some 1,3-Cyclohexadienes to Benzene or Tetralin and Dihydroenophile.
Jacobson, Barry M.,Gerhard, Daniela,Jackson, Cynthia,Smallwood, Joan
, p. 3344 - 3347 (1980)
-
The cyclization of parent and cyclic hexa-1,3-dien-5-ynes - A combined theoretical and experimental study
Prall, Matthias,Krueger, Anke,Schreiner, Peter R.,Hopf, Henning
, p. 4386 - 4394 (2001)
The thermal cycloisomerization of both p...
THE SILICON-MODIFIED METAL AMMONIA REDUCTION OF AROMATIC COMPOUNDS
Rabideau, Peter W.,Karrick, Gregory L.
, p. 2481 - 2484 (1987)
A trimethylsilyl substituent is used to ...
-
Sisido,Nozaki
, (1948)
-
-
Friedman et al.
, p. 1287 (1959)
-
Reduction of polycyclic aromatic hydrocarbons promoted by cobalt or manganese nanoparticles
Nador, Fabiana,Moglie, Yanina,Vitale, Cristian,Yus, Miguel,Alonso, Francisco,Radivoy, Gabriel
, p. 4318 - 4325 (2010)
A new methodology for the partial reduct...
The hydrogenation of aromatic compounds under mild conditions by using a solid Lewis acid and supported palladium catalyst
Zhang, Peng,Wu, Tianbin,Hou, Minqiang,Ma, Jun,Liu, Huizhen,Jiang, Tao,Wang, Weitao,Wu, Congyi,Han, Buxing
, p. 3323 - 3327 (2014)
Al2O3 or SiO2 particles with abundant su...
-
Cerveny,Corson
, p. 2123 (1944)
-
Catalyses of Fe and FeS2 on the Reaction of Di(1-naphthyl)methane
Wei, Xian-yong,Ogata, Eisuke,Niki, Etsuo
, p. 2199 - 2202 (1991)
Reactions of di(1-naphthyl)methane (DNM)...
Swelled plastics in supercritical CO2 as media for stabilization of metal nanoparticles and for catalytic hydrogenation
Ohde, Hiroyuki,Ohde, Mariko,Wai, Chien M.
, p. 930 - 931 (2004)
Swelled plastics in supercritical carbon...
Selective hydrogenation of 1-naphthol on USY-supported NiB nanocatalyst
Wang, Bo,Liu, Hui
, p. 3197 - 3204 (2017)
Selective hydrogenation of 1-naphthol to...
Promotion of Ni/SBA-15 catalyst for hydrogenation of naphthalene by pretreatment with ammonia/water vapour
Ren, Shibiao,Zhang, Ping,Shui, Hengfu,Lei, Zhiping,Wang, Zhicai,Kang, Shigang
, p. 132 - 136 (2010)
The activities of nickel supported on SB...
Water as an additive to enhance the ring opening of naphthalene
Wang, Qian,Fan, Honglei,Wu, Suxiang,Zhang, Zhaofu,Zhang, Peng,Han, Buxing
, p. 1152 - 1158 (2012)
Use of water as a reaction medium or add...
Deep hydrogenation of coal tar over a Ni/ZSM-5 catalyst
Qi, Shi-Chao,Zhang, Lu,Wei, Xian-Yong,Hayashi, Jun-Ichiro,Zong, Zhi-Min,Guo, Lu-Lu
, p. 17105 - 17109 (2014)
We have developed a Ni/ZSM-5 catalyst an...
Hydrodearomatization catalysts based on molybdenum hexacarbonyl Mo(CO)6 supported on mesoporous aromatic frameworks
Maksimov,Karakhanov,Kulikov,Terenina
, p. 589 - 594 (2017)
A method for synthesizing fine hydrodear...
(2,7)TROPONOPHANE
Fujise, Yutaka,Shiokawa, Toshihiro,Mazaki, Yasuhiro,Fukazawa, Yoshimasa,Fujii, Mitsuharu,Ito, Sho
, p. 1601 - 1604 (1982)
The title compound 5 and its dehydro ana...
Iterative Preparation of Platinum Nanoparticles in an Amphiphilic Polymer Matrix: Regulation of Catalytic Activity in Hydrogenation
Hamasaka, Go,Osako, Takao,Srisa, Jakkrit,Torii, Kaoru,Uozumi, Yasuhiro
, p. 147 - 152 (2020)
We demonstrate that iteration of the see...
Preparation of Ni - W aromatic hydrocarbon hydrogenation catalysts by breaking reverse emulsions or suspensions of a precursor in hydrocarbon feedstock
Sizova,Serdyukov,Maksimov
, p. 131 - 137 (2016)
Reverse emulsions (hydrocarbon feedstock...
Nickel–molybdenum sulfide catalysts supported on an ordered mesoporous polymer for hydrogenating–hydrocracking of model biaromatic petroleum compounds
Karakhanov,Boronoev,Ignat’eva,Kardasheva, Yu. S.,Filippova, T. Yu.,Maksimov
, p. 673 - 677 (2017)
Nickel–molybdenum sulfide catalysts have...
-
Wooster,Smith
, p. 179 (1931)
-
Disproportionation of dihydroarenes by sol-gel entrapped RhCl3-quaternary ammonium ion pair catalysts
Rosenfeld, Ayelet,Blum, Jochanan,Avnir, David
, p. 363 - 368 (1996)
The ion pairs generated from RhCl3 · 3H2...
Facile in situ Encapsulation of Highly Dispersed Ni@MCM-41 for the Trans-Decalin Production from Hydrogenation of Naphthalene at Low Temperature
Song, Xiaoyun,Guan, Qingxin,Shu, Yu,Zhang, Xiaojing,Li, Wei
, p. 1286 - 1294 (2019)
Ni@MCM-41 catalyst that has uniformly di...
Naphthalene Hydrogenation over Catalysts Formed In Situ from Ruthenium-Containing Thiosalts
Onishchenko,Suzova,Maximov
, p. 1213 - 1220 (2018)
Abstract: Ruthenium-containing 1-butyl-1...
FeS2-Catalyzed Hydrocracking of Di(1-naphthyl)methane. Effects of Hydrogen Pressure, Catalyst Feed and Reaction Temperature, and Kinetic Study
Wei, Xian-Yong,Ogata, Eisuke,Niki, Etsuo
, p. 987 - 990 (1992)
Hydrocracking of di(1-naphthyl)methane (...
All-Cis Catalytic Hydrogenation of Polynuclear Aromatic Hydrocarbons by Group 5 Metal Aryloxide Compounds
Yu, Joyce S.,Ankianiec, Bernardeta C.,Nguyen, Mindy T.,Rothwell, Ian P.
, p. 1927 - 1929 (1992)
-
MoCx species embedded in ordered mesoporous silica framework with hierarchical structure for hydrogenation of naphthalene
Pang, Min,Chen, Xiaozhen,Xu, Qinying,Liang, Changhai
, p. 146 - 152 (2015)
An integrated route to accomplish the in...
A striking solvent effect on the photochemical reaction of naphthalene with 2-(diethylamino)ethanol
Ogawa,Oh,Satoh,Shirota
, p. 1195 - 1196 (1991)
-
Synthesis of cyclo-1,3-dien-5-ynes
Hopf, Henning,Krueger, Anke
, p. 4378 - 4385 (2001)
Cyclo-1,3-dien-5-ynes with ring sizes fr...
Enhanced selectivity to decalin in naphthalene hydrogenation under supercritical carbon dioxide
Hiyoshi, Norihito,Miura, Ritsuko,Rode, Chandrashekhar V.,Sato, Osamu,Shirai, Masayuki
, p. 424 - 425 (2005)
A charcoal-supported rhodium catalyst wa...
Promotion of hydrogenation activity and sulfur resistance over Ni/ASA catalyst by support modification simultaneously with P and USY
Cui, Sha,Ge, Jiaqi,Wang, Genggeng,Yang, Ying,Liu, Baijun
, p. 41460 - 41470 (2017)
Individual and simultaneous P and USY pr...
Naphthalene hydrogenation over nickel–tungsten sulfide catalysts synthesized in situ from DMSO–hydrocarbon medium emulsions
Sizova,Antonov,Serdyukov,Maksimov
, p. 66 - 70 (2017)
A method for synthesizing unsupported ni...
Chemical treatment of CNTs in acidic KMnO4 solution and promoting effects on the corresponding Pd-Pt/CNTs catalyst
Chen, Jiuling,Chen, Qinghai,Ma, Qing,Li, Yongdan,Zhu, Zhonghua
, p. 114 - 120 (2012)
Carbon nanotubes (CNTs) were chemically ...
Properties of Nanosized Cobalt-Molybdenum Sulfide Catalyst Formed In Situ from Sulfonium Thiosalt
Knyazeva,Panyukova,Maximov
, p. 504 - 510 (2019)
Abstract: A cobalt-molybdenum-containing...
Pyrolysis Studies of Organic Oxygenates. 5. Thermal Chemistry Pathways of 1-Naphthol under Hydrogen
Schlosberg, R. H.,Kurs, A.
, p. 3032 - 3033 (1984)
-
Carbon Dioxide Copolymerization Study with a Sterically Encumbering Naphthalene-Derived Oxide
Darensbourg, Donald J.,Kyran, Samuel J.
, p. 5421 - 5430 (2015)
Poly(1,4-dihydronaphthalene carbonate) h...
Visible Light-Driven, Room Temperature Heck-Type Reaction of Alkyl Halides with Styrene Derivatives Catalyzed by B12 Complex
Chen, Li,Hisaeda, Yoshio,Shimakoshi, Hisashi
, p. 2877 - 2884 (2019)
A visible light driven Heck-type couplin...
Pentacyclo1,6.07,9.08,10>decane: A Cyclopropane Edge-Bridged Prismane and Its Rearrangement to a Fulvene
Kostermans, Gerardus B. M.,Hogenbirk, Marijke,Turkenburg, Lucas A. M.,Wolf, Willem H. de,Bickelhaupt, Friedrich
, p. 2855 - 2857 (1987)
-
From lignocellulosic biomass to renewable cycloalkanes for jet fuels
Zhang, Xuesong,Lei, Hanwu,Zhu, Lei,Wu, Joan,Chen, Shulin
, p. 4736 - 4747 (2015)
A novel pathway was investigated to prod...
(4)PARACYCLOPHANE INTERCEPTED
Kostermans, Gerardus B.M.,Bobeldjik, Marcel,Wolf, Willem H. de,Bickelhaupt, Friedrich
, p. 2471 - 2475 (1987)
Irradiation (254 nm) of 1,4-tetramethyle...
NOVEL POLYHYDRONAPHTHALENIC STRUCTURES
Laguerre, Michel,Dunogues, Jacques,Calas, Raymond
, p. 1227 - 1230 (1981)
Novel 1,7-dihydro- or 1,2,6,7-tetrahydro...
Modular Synthesis of Carbazole-Substituted Phthalimides as Potential Photocatalysts
F?ldesi, Tamás,Gonda, Zsombor,Nagy, Bálint,Novák, Zoltán
supporting information, (2021/11/22)
The modular synthesis of carbazole funct...
Nickel-catalyzed reductive deoxygenation of diverse C-O bond-bearing functional groups
Cook, Adam,MacLean, Haydn,St. Onge, Piers,Newman, Stephen G.
, p. 13337 - 13347 (2021/11/20)
We report a catalytic method for the dir...
Effect of Lanthanum Doping on the reactivity of unsupported CoMoS2 catalysts
Valdes, Carolina,Gonzalez, Diego,Flores, Kenneth,Eubanks,Valle, John,Hernandez, Carlos,Lopez, Jorge,Alcoutlabi, Mataz,Parsons
, (2020/11/24)
In the present study, catalytic systems ...
Selective upgrading of biomass-derived benzylic ketones by (formic acid)–Pd/HPC–NH2 system with high efficiency under ambient conditions
Chen, Yuzhuo,Chen, Zhirong,Gong, Yutong,Mao, Shanjun,Ning, Honghui,Wang, Yong,Wang, Zhenzhen
, p. 3069 - 3084 (2021/11/16)
Upgrading biomass-derived phenolic compo...
119-64-2 Process route
-
-
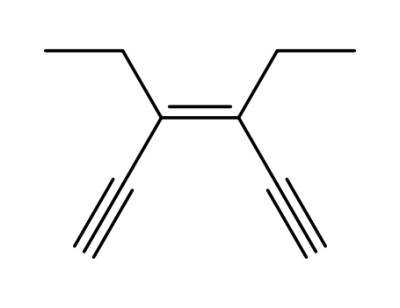
78091-33-5
(Z)-3,4-diethylhexa-1,5-diyn-3-ene

-
-
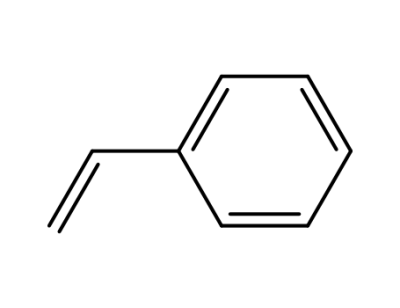
100-42-5,25038-60-2,25247-68-1,28213-80-1,28325-75-9,79637-11-9,9003-53-6
styrene

-
-
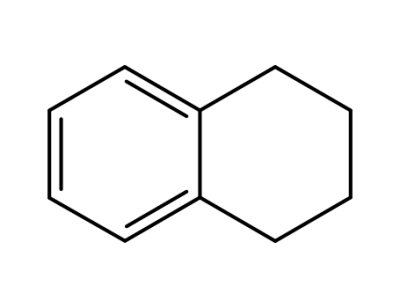
119-64-2
tetralin

-
-
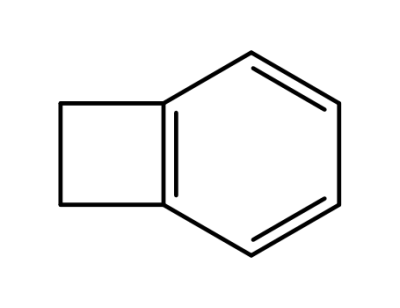
694-87-1
benzocyclobutene

-
-
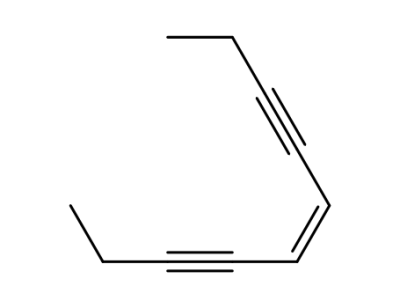
122911-16-4
cis-5-decene-3,7-diyne
| Conditions | Yield |
|---|---|
|
at 500 ℃;
Further byproducts given;
|
76.8% 7.8% 1.3% 1.6% |
-
-
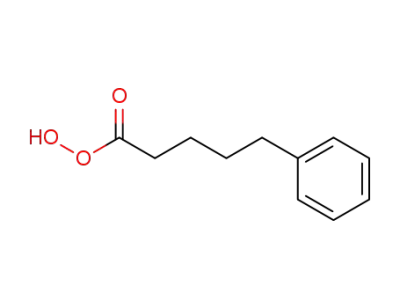
85696-63-5
peracide phenyl-5 pentanoique

-

-
119-64-2
tetralin

-
-
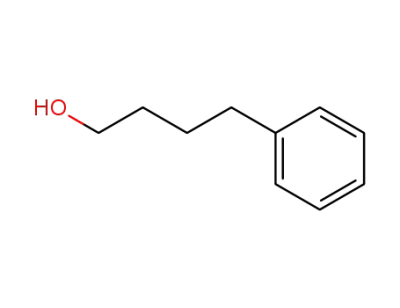
3360-41-6
4-phenyl-butan-1-ol

-
-
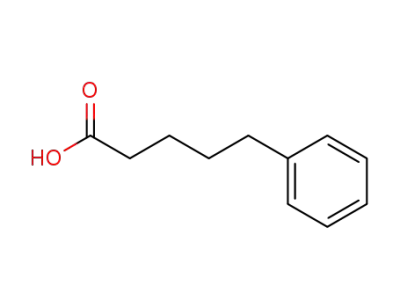
2270-20-4
5-Phenylpentanoic acid

-
-
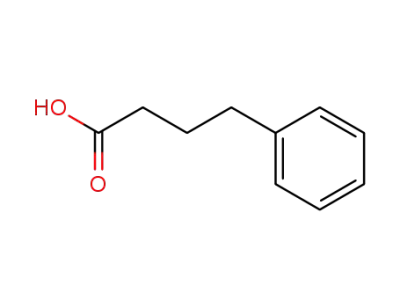
1821-12-1
4-Phenylbutyric acid
| Conditions | Yield |
|---|---|
|
In
n-heptane;
at 98 ℃;
Further byproducts given;
|
75 % Chromat. 4 % Chromat. 7 % Chromat. 2 % Chromat. |
119-64-2 Upstream products
-
2461-34-9
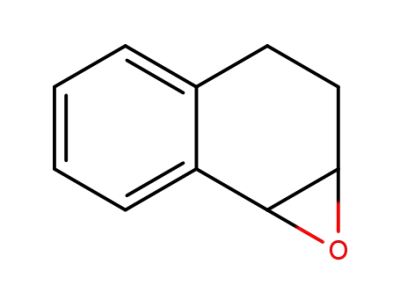
1a,2,3,7b-tetrahydronaphtho[1,2-b]oxirene
-
91-20-3
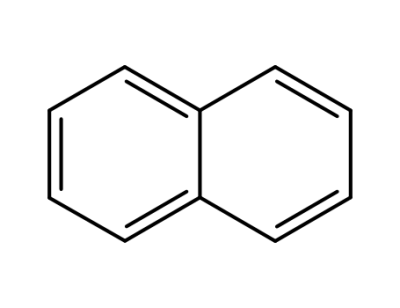
naphthalene
-
90-15-3
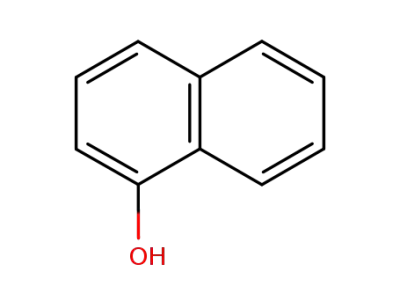
α-naphthol
-
447-53-0
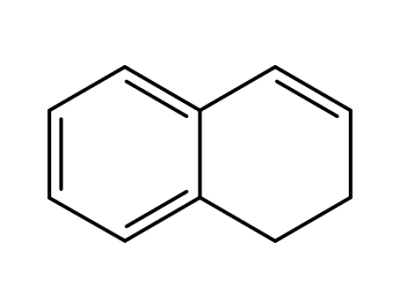
1,2-Dihydronaphthalene
119-64-2 Downstream products
-
466-95-5
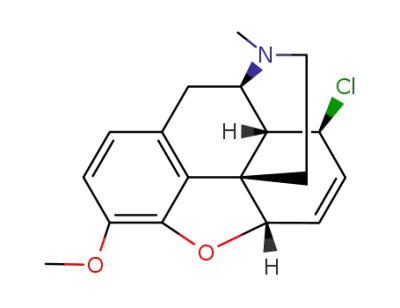
8β-chloro-4,5α-epoxy-3-methoxy-17-methyl-morphin-6-ene
-
91-20-3

naphthalene
-
75-07-0
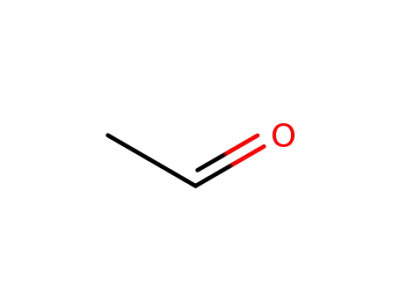
acetaldehyde
-
108-88-3
toluene
Relevant Products
-
DIBASIC ESTER
CAS:95481-62-2
-
1H,1H,2H,2H-Perfluorodecyltrimethoxysilane
CAS:83048-65-1
-
1H,1H,2H,2H-Perfluorooctyltrichlorosilane
CAS:78560-45-9