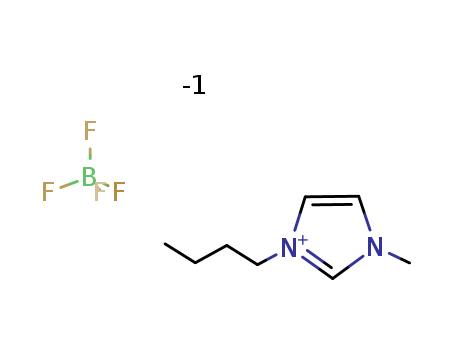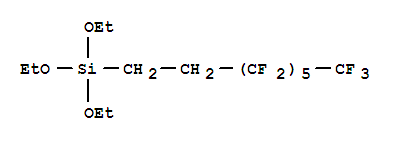
429-42-5
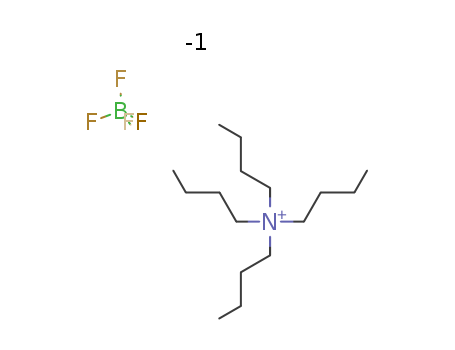
- Product Name:Tetrabutylammonium tetrafluoroborate
- Molecular Formula:C16H36BF4N
- Purity:99%
- Molecular Weight:329.273
Product Details;
CasNo: 429-42-5
Molecular Formula: C16H36BF4N
Appearance: white crystalline powder
factory and manufacture 429-42-5 Tetrabutylammonium tetrafluoroborate lonic liquid
- Molecular Formula:C16H36BF4N
- Molecular Weight:329.273
- Appearance/Colour:white crystalline powder
- Melting Point:155-161 °C
- Flash Point:>93℃
- PSA:0.00000
- LogP:6.30360
Tetrabutylammonium tetrafluoroborate(Cas 429-42-5) Usage
|
Purification Methods |
Recrystallise it from H2O, aqueous EtOH or from EtOAc by cooling in Dry-ice. Also recrystallise it from ethyl acetate/pentane or dry acetonitrile. Dry it at 80o under vacuum. [Detty & Jones J Am Chem Soc 109 5666 1987, Hartley & Faulkner J Am Chem Soc 107 3436 1985.] The acetate has m 118±2o (from BuCl), the bromide has m 118o (from EtOAc) and the nitrate has m 120o (from *C6H6). [Witschonka & Kraus J Am Chem Soc 69 2472 1947, Wheeler & Sandstedt J Am Chem Soc 77 2024 1955, Beilstein 4 IV 558.] |
|
General Description |
Tetrabutylammonium tetrafluoroborate (TBATFB) is a phase transfer catalyst. It can be synthesized by the reaction between 30% aqueous solution of tetrafluoroboric acid and 40% aqueous solution of tetrabutylamonium hydroxide. Tetrabutylammonium tetrafluoroborate acts as an electrolyte and inhibits the self-assembly of alkylthiosulfate on gold. |
InChI:InChI=1/C16H36N.BF4/c1-5-9-13-17(14-10-6-2,15-11-7-3)16-12-8-4;2-1(3,4)5/h5-16H2,1-4H3;/q+1;-1
429-42-5 Relevant articles
Electrochemical studies of the nickel catecholate complexes for detection of sulphur dioxide gas
Tembwe, Inonge,Ngila, J. Catherine,Kgarebe, Boitumelo,Darkwa, James,Iwuoha, Emmanuel
, p. 4314 - 4318 (2010)
Nickel catecholate complexes, bis(diphen...
External oxidant-free cross-coupling: Electrochemically induced aromatic C-H phosphonation of azoles with dialkyl-: H -phosphonates under silver catalysis
Yurko,Gryaznova,Kholin,Khrizanforova,Budnikova
, p. 190 - 196 (2018)
A convenient external oxidant-free metho...
Controlling Metal-to-Oxygen Ratios via M=O Bond Cleavage in Polyoxovanadate Alkoxide Clusters
Petel, Brittney E.,Fertig, Alex A.,Maiola, Michela L.,Brennessel, William W.,Matson, Ellen M.
, p. 10462 - 10471 (2019)
In this manuscript, we further investiga...
Halogen-free ionic liquids: Effect of chelated orthoborate anion structure on their lubrication properties
Gusain, Rashi,Khatri, Om P.
, p. 25287 - 25294 (2015)
In transportation vehicles, a large port...
Assessing the Electrocatalytic Properties of the (Cp*RhIII)2+-Polyoxometalate Derivative [H2PW11O39(RhIIICp*(OH2))]3- towards CO2 Reduction
Girardi, Marcelo,Platzer, Dominique,Griveau, Sophie,Bedioui, Fethi,Alves, Sandra,Proust, Anna,Blanchard, Sébastien
, (2018)
Storage of electricity produced intermit...
Transition-metal Complexes of Crown Ether Benzodithiolenes. Part 2. The Effects of Alkali-metal Cation Binding
Lowe, Nigel D.,Garner, C. David
, p. 3333 - 3340 (1993)
The binding of Li(+), Na(+) and K(+) cat...
Stereoelectronic and Resonance Effects on the Rate of Ring Opening of N-Cyclopropyl-Based Single Electron Transfer Probes
Grimm, Michelle L.,Suleman, N. Kamrudin,Hancock, Amber N.,Spencer, Jared N.,Dudding, Travis,Rowshanpour, Rozhin,Castagnoli, Neal,Tanko, James M.
supporting information, p. 2640 - 2652 (2020/02/18)
N-Cyclopropyl-N-methylaniline (5) is a p...
Charge-Assisted phosph(v)azane anion receptors
Bond, Andrew D.,Goodman, Jonathan M.,Lee, Sanha,Plajer, Alex J.,Wright, Dominic S.
, p. 3403 - 3407 (2020/04/02)
Coordination of Cu(i) or Pd(ii) to selen...
Lewis Acidity Scale of Diaryliodonium Ions toward Oxygen, Nitrogen, and Halogen Lewis Bases
Legault, Claude Y.,Mayer, Robert J.,Mayr, Herbert,Ofial, Armin R.
supporting information, (2020/03/13)
Equilibrium constants for the associatio...
429-42-5 Process route
-
-
1375484-96-0
[Bu4N][C4H9BF3]

-
-
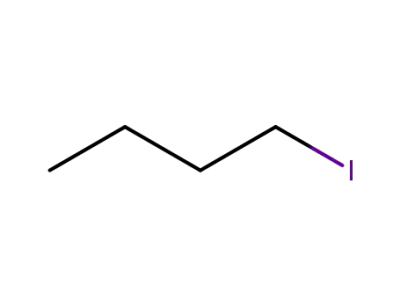
542-69-8
1-iodo-butane

-
-
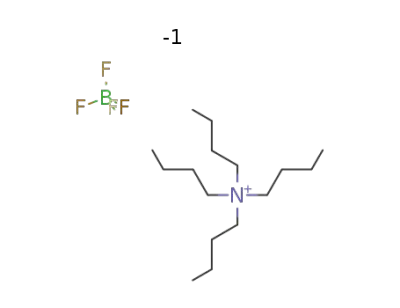
429-42-5
tetrabutylammonium tetrafluoroborate

-
-
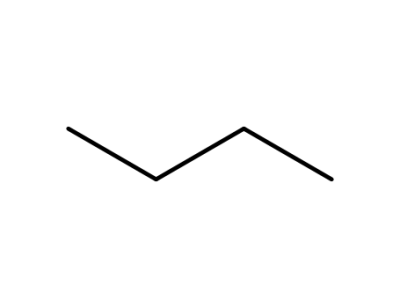
106-97-8,9003-29-6,9021-92-5
n-butane
| Conditions | Yield |
|---|---|
|
With
Iodine monochloride;
In
dichloromethane;
at 20 ℃;
for 1h;
|
-
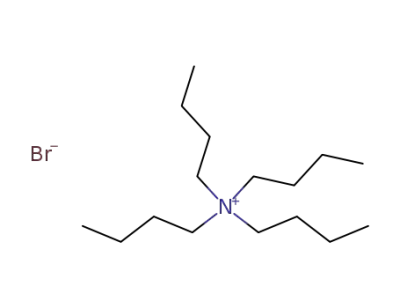
-
1643-19-2
tetrabutylammomium bromide

-

-
429-42-5
tetrabutylammonium tetrafluoroborate
| Conditions | Yield |
|---|---|
|
With
potassium tetrafluoroborate;
In
dichloromethane; water;
at 20 ℃;
for 24h;
|
96% |
|
With
potassium tetrafluoroborate;
In
dichloromethane; water;
at 20 ℃;
for 24h;
|
96% |
|
With
potassium tetrafluoroborate;
In
dichloromethane; water;
at 20 ℃;
for 24h;
|
96% |
|
With
trimethyl phosphite; tetrafluoroboric acid;
at 0 - 60 ℃;
for 15h;
Inert atmosphere;
neat (no solvent);
|
90% |
|
With
potassium tetrafluoroborate;
In
water;
|
|
|
With
sodium tetrafluoroborate;
In
water;
|
429-42-5 Upstream products
-
10534-59-5
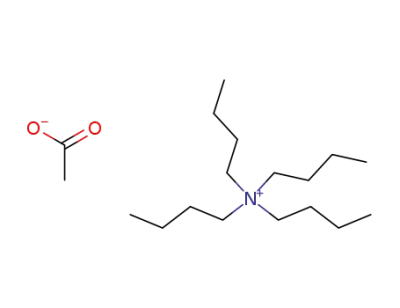
tetrabutylammonium acetate
-
49723-76-4
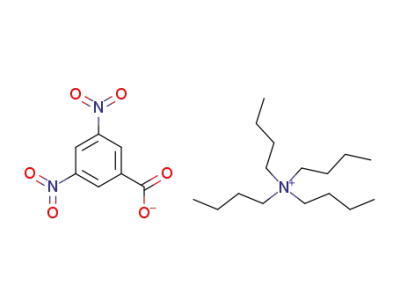
tetrabutylammonium 3,5-dinitrobenzoate
-
60931-59-1
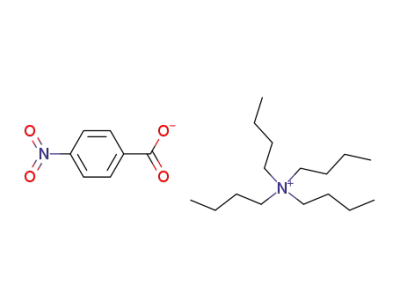
tetrabutylammonium 4-nitrobenzoate
429-42-5 Downstream products
-
102-82-9

tributyl-amine
-
2752-95-6
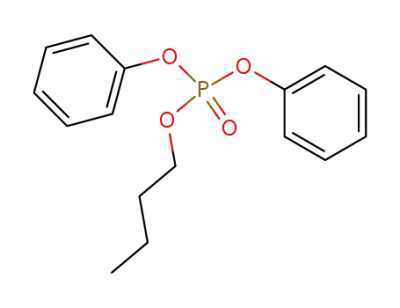
butyl diphenyl phosphate
-
46438-39-5
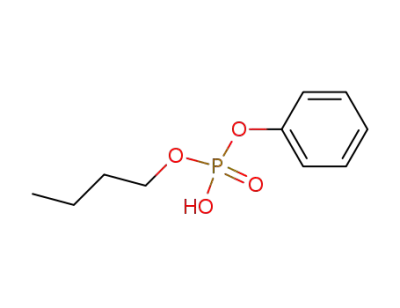
Phosphoric Acid Butyl Phenyl Ester
-
71-43-2
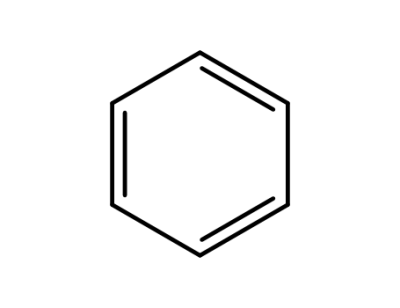
benzene
Relevant Products
-
1-Butyl-3-methylimidazolium tetrafluoroborate
CAS:174501-65-6
-
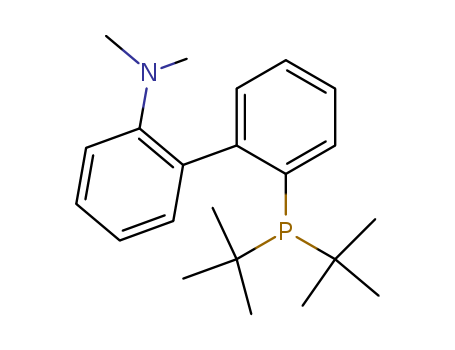
2-Di-tert-butylphosphino-2'-(N,N-dimethylamino)biphenyl
CAS:224311-49-3
-
Trifluorooctyl triethoxy silane
CAS:51851-37-7