50824-05-0
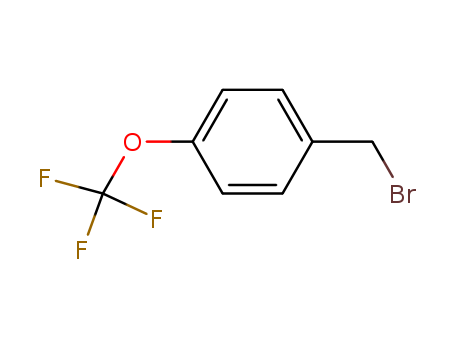
- Product Name:4-(Trifluoromethoxy)Benzyl Bromide
- Molecular Formula:C8H6OBrF3
- Purity:99%
- Molecular Weight:255.034
Product Details;
CasNo: 50824-05-0
Molecular Formula: C8H6OBrF3
Appearance: yellow or slight yellow liquid
factory and supplier 50824-05-0 4-(Trifluoromethoxy)Benzyl Bromide in stock
- Molecular Formula:C8H6OBrF3
- Molecular Weight:255.034
- Appearance/Colour:yellow or slight yellow liquid
- Vapor Pressure:3.44mmHg at 25°C
- Melting Point:22-24 °C
- Refractive Index:n20/D 1.48(lit.)
- Boiling Point:203.2 °C at 760 mmHg
- Flash Point:94.4 °C
- PSA:9.23000
- Density:1.6 g/cm3
- LogP:3.48010
4-(Trifluoromethoxy)benzyl bromide(Cas 50824-05-0) Usage
|
General Description |
Polymerizations of 4-(trifluoromethoxy)benzyl bromide, via Friedel-Crafts polymerization using aluminum chloride as a catalyst has been reported. |
InChI:InChI=1/C9H7F9O2/c1-2-5(19)20-4-3-6(10,11)7(12,8(13,14)15)9(16,17)18/h2H,1,3-4H2
50824-05-0 Relevant articles
Synthesis and biological evaluation of curcumin inspired indole analogues as tubulin polymerization inhibitors
Sri Ramya,Angapelly, Srinivas,Guntuku, Lalita,Singh Digwal, Chander,Nagendra Babu, Bathini,Naidu,Kamal, Ahmed
, p. 100 - 114 (2016/12/30)
In our endeavour towards the development...
Iron-catalyzed borylation of alkyl, allyl, and aryl halides: Isolation of an iron(I) boryl complex
Bedford, Robin B.,Brenner, Peter B.,Carter, Emma,Gallagher, Timothy,Murphy, Damien M.,Pye, Dominic R.
supporting information, p. 5940 - 5943 (2015/01/08)
Activation of B2pin2 with tBuLi facilita...
SULFONYL-SUBSTITUTED BICYCLIC COMPOUNDS AS MODULATORS OF PPAR
-
Page/Page column 17, (2008/06/13)
The present invention relates to compoun...
Further naphthylcombretastatins. An investigation on the role of the naphthalene moiety
Maya, Ana B. S.,Pérez-Melero, Concepción,Mateo, Carmen,Alonso, Dulce,Fernández, José Luis,Gajate, Consuelo,Mollinedo, Faustino,Peláez, Rafael,Caballero, Esther,Medarde, Manuel
, p. 556 - 568 (2007/10/03)
By synthesis and biological studies of n...
50824-05-0 Process route
-
-
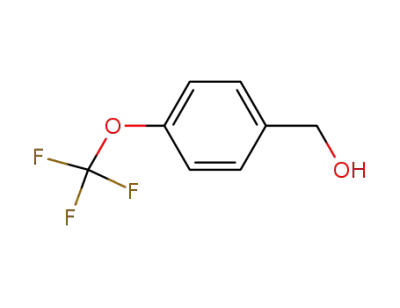
1736-74-9
4-trifluoromethoxybenzyl alcohol

-
-
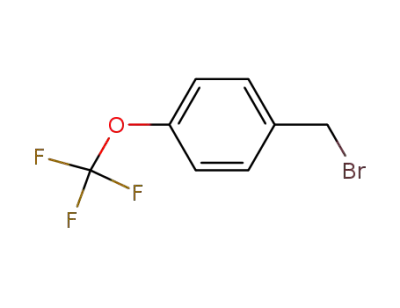
50824-05-0
4-trifluoromethoxybenzyl bromide
| Conditions | Yield |
|---|---|
|
With
phosphorus tribromide;
In
dichloromethane;
at 0 - 20 ℃;
for 3.5h;
|
66% |
|
With
hydrogen bromide;
|
|
|
With
phosphorus tribromide;
In
diethyl ether;
at -40 ℃;
|
|
|
With
N-Bromosuccinimide; triphenylphosphine;
In
dichloromethane;
at 0 - 20 ℃;
for 3h;
|
|
|
With
phosphorus tribromide;
In
diethyl ether;
at 0 ℃;
for 0.5h;
|
-
-
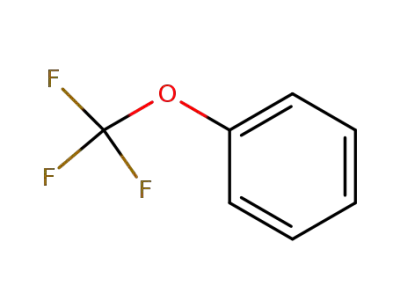
456-55-3
1-trifluoromethoxybenzene

-

-
50824-05-0
4-trifluoromethoxybenzyl bromide
| Conditions | Yield |
|---|---|
|
With
sodium bromide; sulfuric acid; paraformaldehyde;
In
tert-butyl methyl ether; water; acetic acid;
|
60% |
50824-05-0 Upstream products
-
1736-74-9

4-trifluoromethoxybenzyl alcohol
-
659-28-9
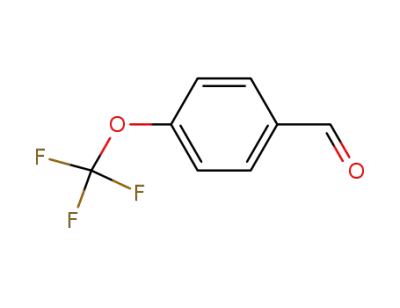
p-trifluoromethoxybenzaldehyde
-
587-18-8
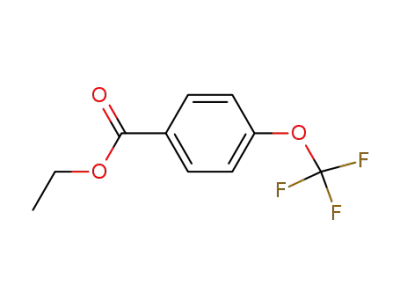
4-trifluoromethoxy-benzoic acid ethyl ester
-
456-55-3

1-trifluoromethoxybenzene
50824-05-0 Downstream products
-
340759-27-5
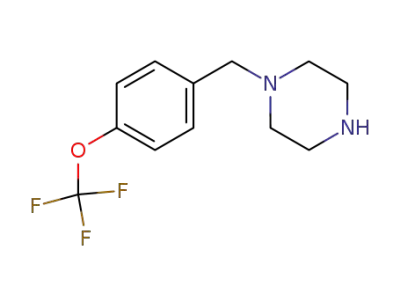
1-(4-(trifluoromethoxy)benzyl)piperazine
-
280135-08-2
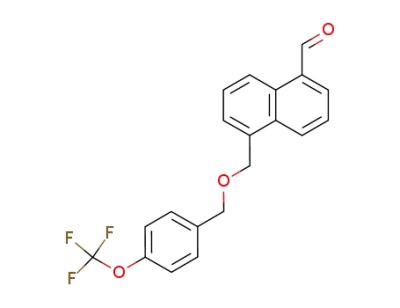
5-(4-trifluoromethoxybenzyloxy)methyl-1-naphthaldehyde
-
616882-14-5
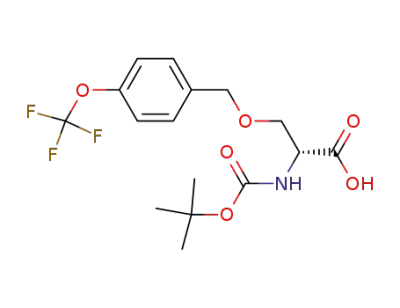
N-tert-butoxycarbonylamino-O-(4-trifluoromethoxybenzyl)-D-serine
Relevant Products
-
5-Bromo-2-(trifluoromethoxy)aniline
CAS:886762-08-9
-
1-Butyl-2,3-Dimethylimidazolium Bromide
CAS:475575-45-2
-
2-Bromobenzotrifluoride
CAS:392-83-6