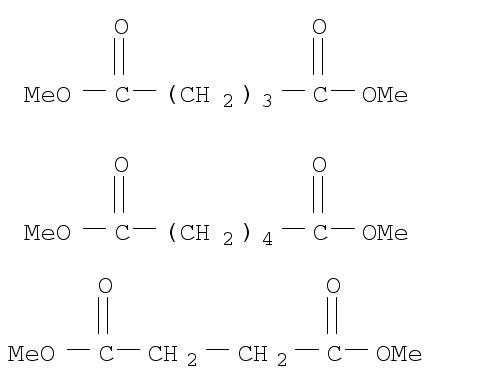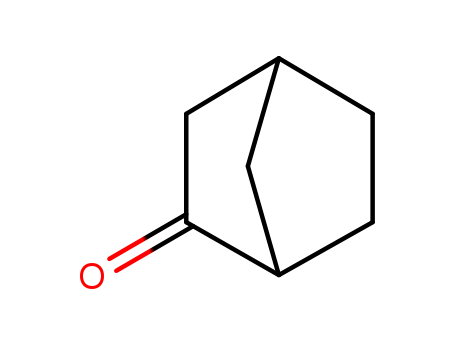
497-38-1
- Product Name:2-Norbornanone
- Molecular Formula:C7H10 O
- Purity:99%
- Molecular Weight:110.156
Product Details;
CasNo: 497-38-1
Molecular Formula: C7H10 O
Appearance: colorless to white adhering crystals
factory and supplier 497-38-1 2-Norbornanone in stock
- Molecular Formula:C7H10 O
- Molecular Weight:110.156
- Appearance/Colour:colorless to white adhering crystals
- Vapor Pressure:1.5mmHg at 25°C
- Melting Point:93-96ºC(lit.)
- Refractive Index:1.51
- Boiling Point:168-172 ºC(lit.)
- Flash Point:93 ºF
- PSA:17.07000
- Density:1.082g/cm3
- LogP:1.37550
NORCAMPHOR(Cas 497-38-1) Usage
|
Purification Methods |
Crystallise it from water and sublime it in vacuo. It has at max 287nm (EtOH). The semicarbazone has m 196-196.5o (from EtOH/H2O). The 2,4-dinitrophenylhydrazone has m 137-138o (from EtOH). [Wildman & Hemminger J Org Chem 17 1641 1952, Wood & Roberts J Org Chem 23 1124 1957, Bixter & Niemann J Org Chem 23 742 1958, Beilstein 7 III 243, 7 IV 139.] |
InChI:InChI=1/C7H10O/c8-7-4-5-1-2-6(7)3-5/h5-6H,1-4H2/t5-,6-/m1/s1
497-38-1 Relevant articles
Acid-catalyzed hydrolysis of bridged bi- and tricyclic compounds. XXXVII. Kinetics and mechanisms of 1- and 3-acetoxynortricyclanes
Lajunen, Martti,Yli-Mannila, Rita,Salminen, Outi
, p. 133 - 138 (2000)
The disappearance of 3- and 1-acetoxynor...
Synthetic Methods and Reactions; 77. Dimethyl Sulfoxide/Chlorosulfonyl Isocyanate: An Extremely Mild Reagent for Oxidation of Alcohols to Carbonyl Compounds
Olah, George A.,Vankar, Yashwant D.,Arvanaghi, Massoud
, p. 141 - 142 (1980)
-
Role of Dioxygen as an Activator in Olefin Metathesis
Ivin, Kenneth J.,Reddy, Boreddy S. R.,Rooney, John J.
, p. 1062 - 1064 (1981)
Evidence is presented that the beneficia...
Acid-catalyzed hydrolysis of bridged bi- and tricyclic compounds. XXXVIII - Kinetics and mechanisms of 1- and 3-nortricyclanols
Lajunen, Martti,Lahti, Veli
, p. 854 - 858 (2001)
The disappearance of 1- and 3-nortricycl...
OXIDATION OF NON-ACTIVATED C-H BONDS IN HYDROCARBONS AND STEROIDS
Linz, T.,Schaefer, H. J.
, p. 6581 - 6582 (1987)
CrO3 in CH2Cl2/CH3COOH/(CH3CO)2O oxidize...
Supported rhenium nanoparticle catalysts for acceptorless dehydrogenation of alcohols: Structure-activity relationship and mechanistic studies
Kon, Kenichi,Onodera, Wataru,Toyao, Takashi,Shimizu, Ken-Ichi
, p. 5864 - 5870 (2016)
Al2O3-supported Re with different oxidat...
Improved synthesis of 2-norbornanone
Zhang, Heng,Fu, Shuixiang,Tao, Ming,Liao, Zutai,Qian, Chao,Chen, Xinzhi
, p. 3325 - 3332 (2016)
Herein a two-step process for the synthe...
Liquid-phase oxidation of olefins with rare hydronium ion salt of dinuclear dioxido-vanadium(V) complexes and comparative catalytic studies with analogous copper complexes
Maurya, Abhishek,Haldar, Chanchal
, (2021/02/26)
Homogeneous liquid-phase oxidation of a ...
New Cu(II), Co(II) and Ni(II) azo-Schiff base complexes: Synthesis, characterization, catalytic oxidation of alkenes and DFT study
Lashanizadegan, Maryam,Asna Ashari, Hadiseh,Sarkheil, Marzieh,Anafcheh, Maryam,Jahangiry, Samira
, (2021/03/22)
Three new complexes with general formula...
A sustainable approach towards solventless organic oxidations catalyzed by polymer immobilized Nb(V)-peroxido compounds with H2O2 as oxidant
Ahmed, Kabirun,Gogoi, Sandhya Rani,Islam, Nashreen S.,Saikia, Gangutri,Sultana, Sazida Yasmin,Talukdar, Hiya
, (2021/11/16)
New heterogeneous catalysts comprising o...
Reduced Amino Acid Schiff Base-Iron(III) Complexes Catalyzing Oxidation of Cyclohexane with Hydrogen Peroxide
Zheng, Anna,Zhou, Qingqing,Ding, Bingjie,Li, Difan,Zhang, Tong,Hou, Zhenshan
, p. 3385 - 3395 (2021/08/23)
The reduced amino acid Schiff base ligan...
497-38-1 Process route
-
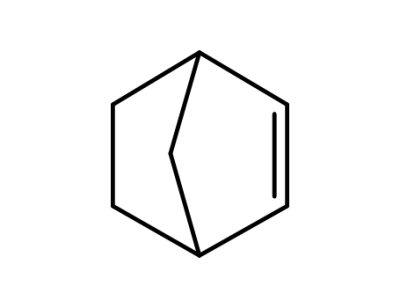
-
498-66-8
norborn-2-ene

-
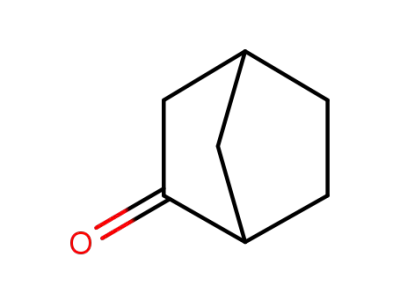
-
497-38-1
Norbornan-2-on

-
-
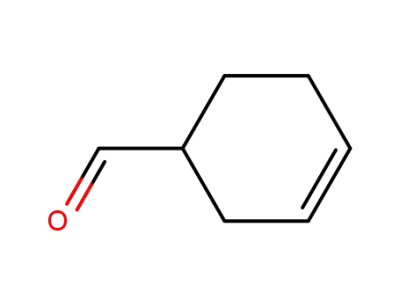
100-50-5,61746-36-9
3-Cyclohexene-1-carboxaldehyde

-
-
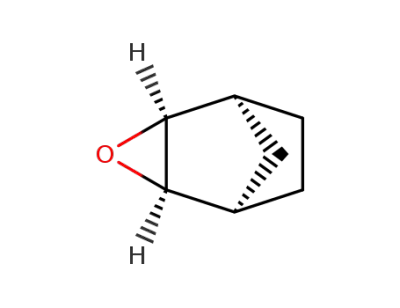
278-74-0,3146-39-2,57378-36-6
exo-2,3-epoxynorbornane

-
-
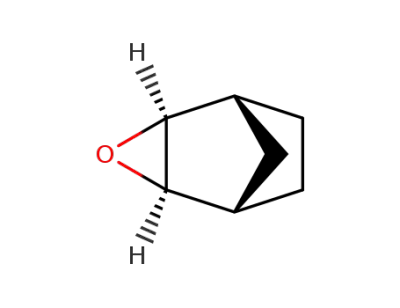
844874-18-6
exo-2,3-epoxynorbornane
| Conditions | Yield |
|---|---|
|
With
iodosylbenzene;
TFTEP*FeCl;
In
dichloromethane;
at 0 ℃;
Yields of byproduct given;
|
3.9% 0.3% |
|
With
iodosylbenzene;
OEP*FeCl;
In
dichloromethane;
at 0 ℃;
Yield given. Yields of byproduct given;
|
1.6% 0.2% |
|
With
iodosylbenzene;
TFTEP*MnCl;
In
dichloromethane;
at 0 ℃;
Yield given. Yields of byproduct given;
|
0.8% 0.2% |
|
With
iodosylbenzene;
TFTEP*MnCl;
In
dichloromethane;
at 0 ℃;
Product distribution;
other catalysts; other olefines;
|
0.8% 0.2% |
|
With
1,2,3,4,5-pentafluoro-6-iodobenzene;
Fe(2,6-Cl)PPCl;
In
dichloromethane;
Product distribution;
Mechanism;
Ambient temperature;
further complexes of various tetraphenylporphyrins with various metals;
|
|
|
With
[VO(OMe)((E)-3-hydroxy-N'-(2-hydroxybenzylidene)2-2naphthohydrazide(-2H))]; dihydrogen peroxide;
In
water; acetonitrile;
at 60 ℃;
for 4h;
under 760.051 Torr;
|
67 %Chromat. 14 %Chromat. 8 %Chromat. 8 %Chromat. |
-

-
498-66-8
norborn-2-ene

-

-
497-38-1
Norbornan-2-on

-

-
100-50-5,61746-36-9
3-Cyclohexene-1-carboxaldehyde

-

-
844874-18-6
exo-2,3-epoxynorbornane
| Conditions | Yield |
|---|---|
|
With
pyrimido<5.4-g>pteridine 10-oxide;
In
acetonitrile;
Product distribution;
Mechanism;
Ambient temperature;
Irradiation;
|
42 % Chromat. 4 % Chromat. |
497-38-1 Upstream products
-
623-11-0
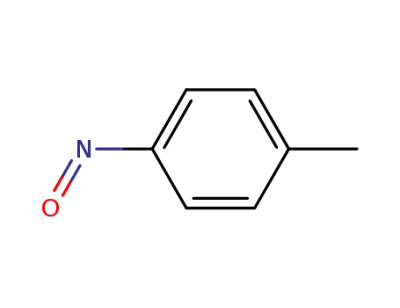
1-methyl-4-nitrosobenzene
-
6343-65-3
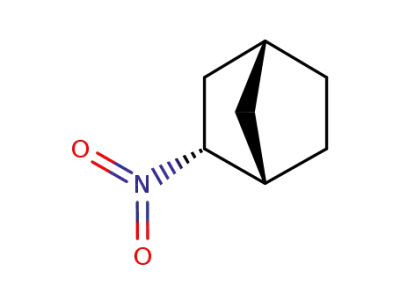
endo-2-nitronorbornane
-
497-36-9
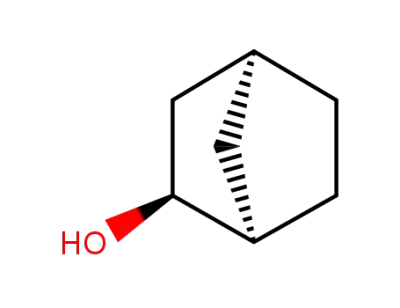
endo-norborneol
-
408325-53-1
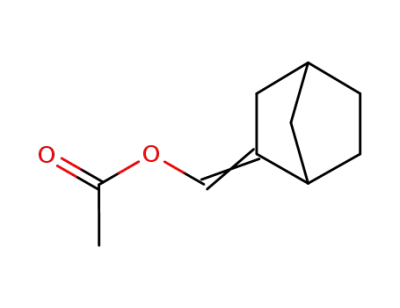
acetic acid-[2]norbornylidenemethyl ester
497-38-1 Downstream products
-
6236-71-1
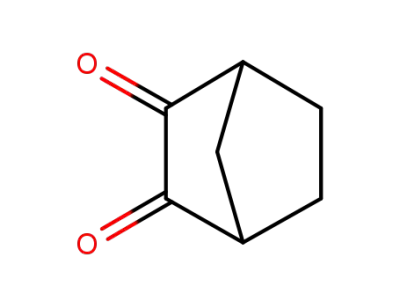
bicyclo<2.2.1>heptane-2,3-dione
-
53600-88-7
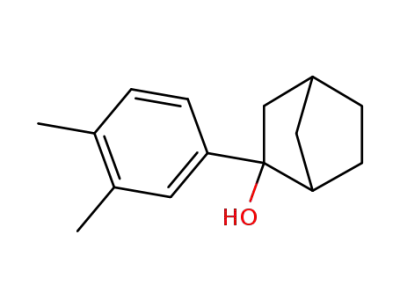
2-(3,4-Dimethyl-phenyl)-bicyclo[2.2.1]heptan-2-ol
-
78195-95-6
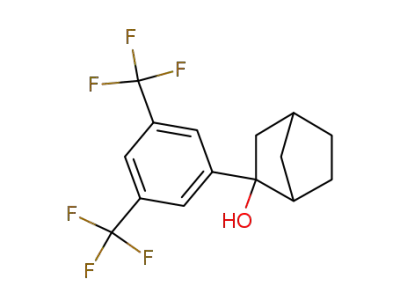
2-(3,5-Bis-trifluoromethyl-phenyl)-bicyclo[2.2.1]heptan-2-ol
-
52329-81-4
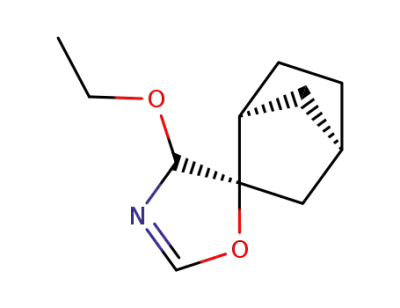
4'ξ-ethoxy-4'H-(2endoO,5'rC1)-spiro[bicyclo[2.2.1]heptane-2,5'-oxazole]
Relevant Products
-
DIBASIC ESTER
CAS:95481-62-2
-
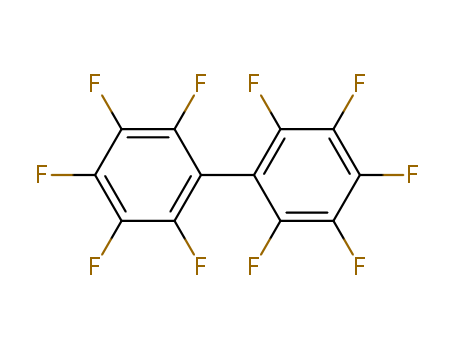
Octafluorotoluene
CAS:434-64-0
-
5-Norbornene-2-methanol
CAS:95-12-2